Respiratory Pathology — MCQs

On this page
In ARDS, which cells are primarily damaged?
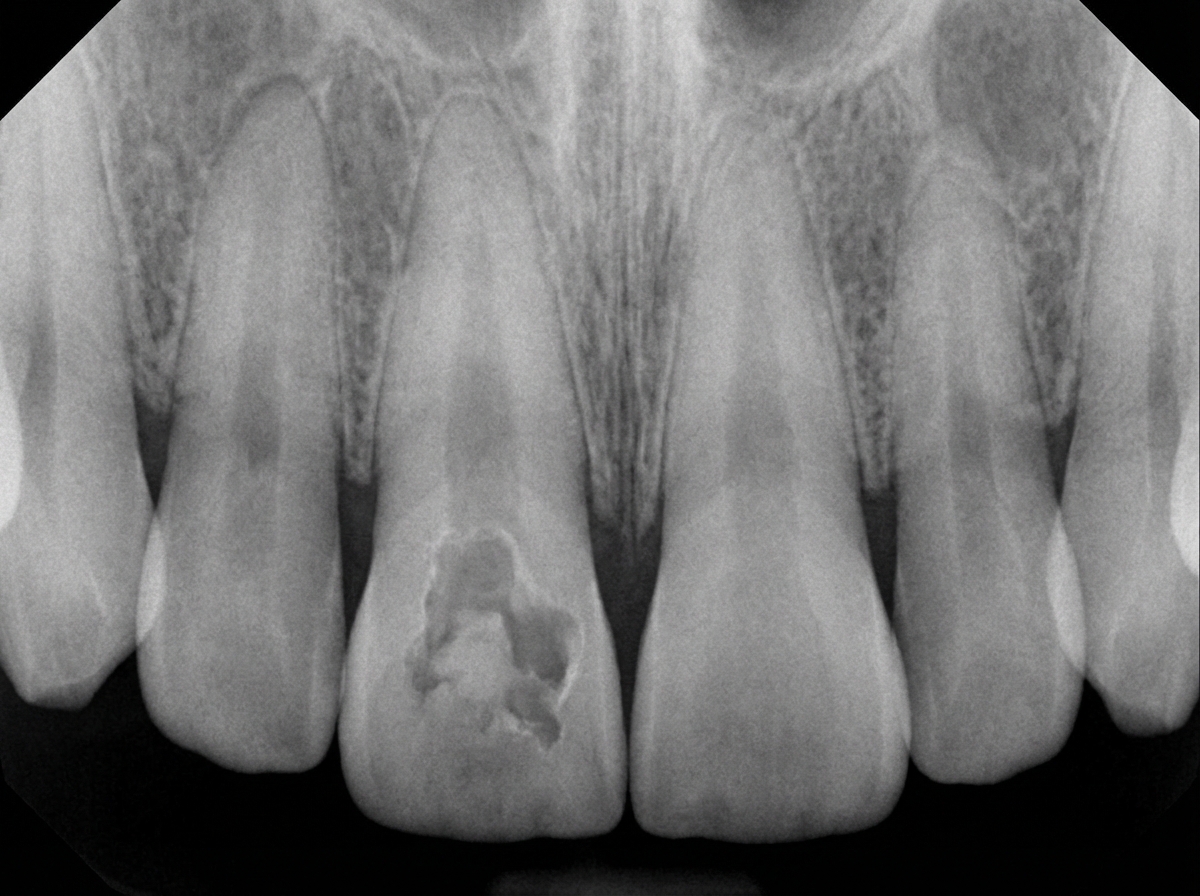
A patient gives a history of previous chronic abscess on the deciduous precursor of tooth 11. The following radiograph indicates:
Dentigerous cysts originate from which structure?
Which of the following is a variant of bronchoalveolar carcinoma?
Which of the following substances is NOT secreted by the bronchial mucosa?
What is the most common lung malformation?
A scar in lung tissue may transform into which type of carcinoma?
A 65-year-old male diagnosed by biopsy with squamous cell carcinoma of the lung presents with very high serum calcium levels. What is the probable cause of hypercalcemia?
Lung carcinoma is most commonly associated with which of the following?
The dangerous particle size causing pneumoconiosis varies from:
Practice by Chapter
Congenital Anomalies
Practice Questions
Atelectasis and Acute Lung Injury
Practice Questions
Obstructive Pulmonary Diseases
Practice Questions
Restrictive Pulmonary Diseases
Practice Questions
Lung Infections
Practice Questions
Pulmonary Vascular Diseases
Practice Questions
Lung Tumors
Practice Questions
Pleural Diseases
Practice Questions
Interstitial Lung Diseases
Practice Questions
Occupational Lung Diseases
Practice Questions
Want unlimited practice?
Get full access to all questions, explanations, and performance tracking.
Scan to download app
