Respiratory Pathology — MCQs

On this page
Which of the following pulmonary conditions is associated with the widespread formation of hyaline membranes in the alveolar cavities?
In centrilobular (centriacinar) emphysema, there is an abnormal, permanent enlargement of which structure?
Which of the following is NOT seen in a Pancoast tumor?
What are heart failure cells?
All of the following statements are true regarding silicosis, except?
Smoking is most strongly associated with which type of lung cancer?
The histologic entity that has the worst prognosis in interstitial lung disease is?
A 45-year-old man with a 15-year history of working in a construction factory presents with a 6-month history of cough. What is the probable diagnosis based on his chest X-ray findings and occupational history?
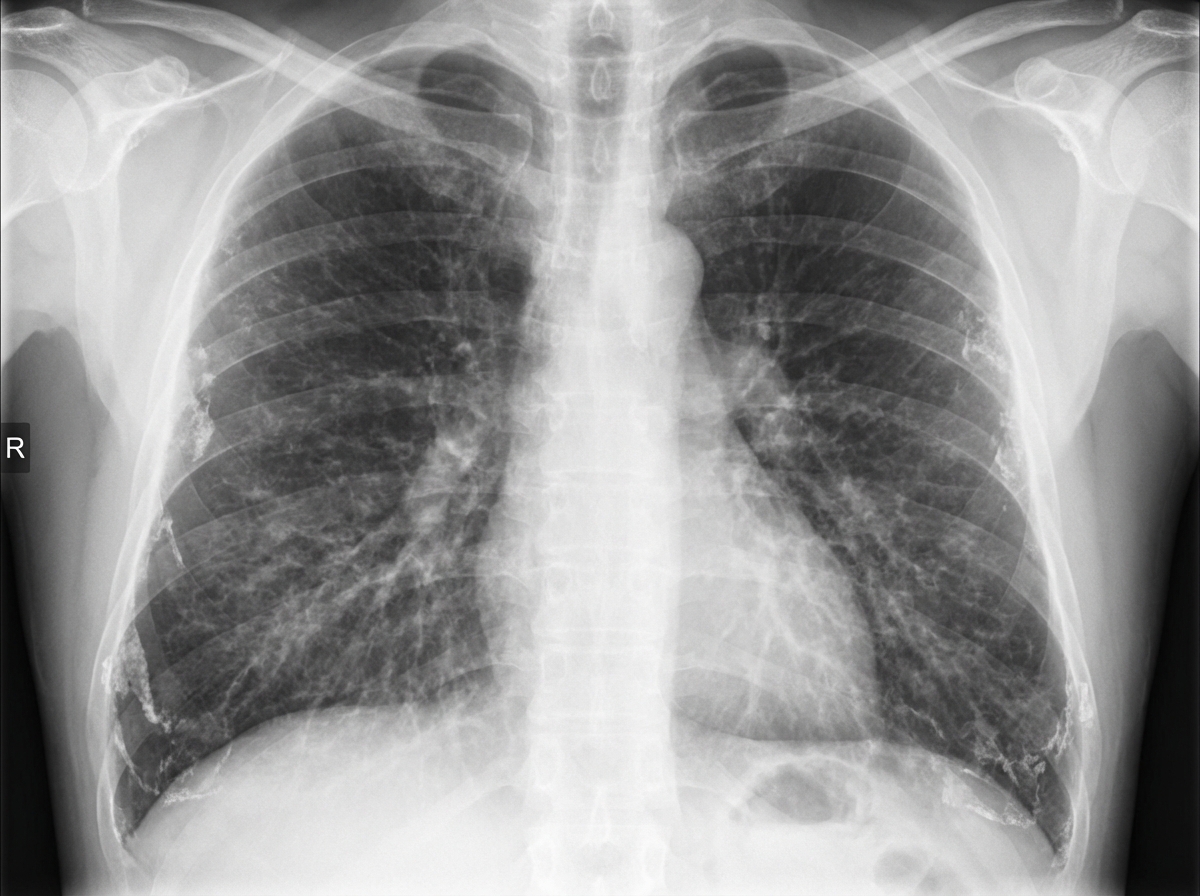
What is the most common type of emphysema associated with alpha-1 antitrypsin deficiency?
Bilateral lymphadenopathy, along with non-caseating granulomas, is a characteristic feature of which condition?
Practice by Chapter
Congenital Anomalies
Practice Questions
Atelectasis and Acute Lung Injury
Practice Questions
Obstructive Pulmonary Diseases
Practice Questions
Restrictive Pulmonary Diseases
Practice Questions
Lung Infections
Practice Questions
Pulmonary Vascular Diseases
Practice Questions
Lung Tumors
Practice Questions
Pleural Diseases
Practice Questions
Interstitial Lung Diseases
Practice Questions
Occupational Lung Diseases
Practice Questions
Want unlimited practice?
Get full access to all questions, explanations, and performance tracking.
Scan to download app
