Reproductive Pathology — MCQs

On this page
Oestrogen causes proliferation of which cells in the vaginal epithelium?
Which of the following is NOT true about spermatocytic seminoma?
What is the most common testicular tumor in the prepubertal age group?
What is the interpretation of the given cervix biopsy of a 50-year-old lady?
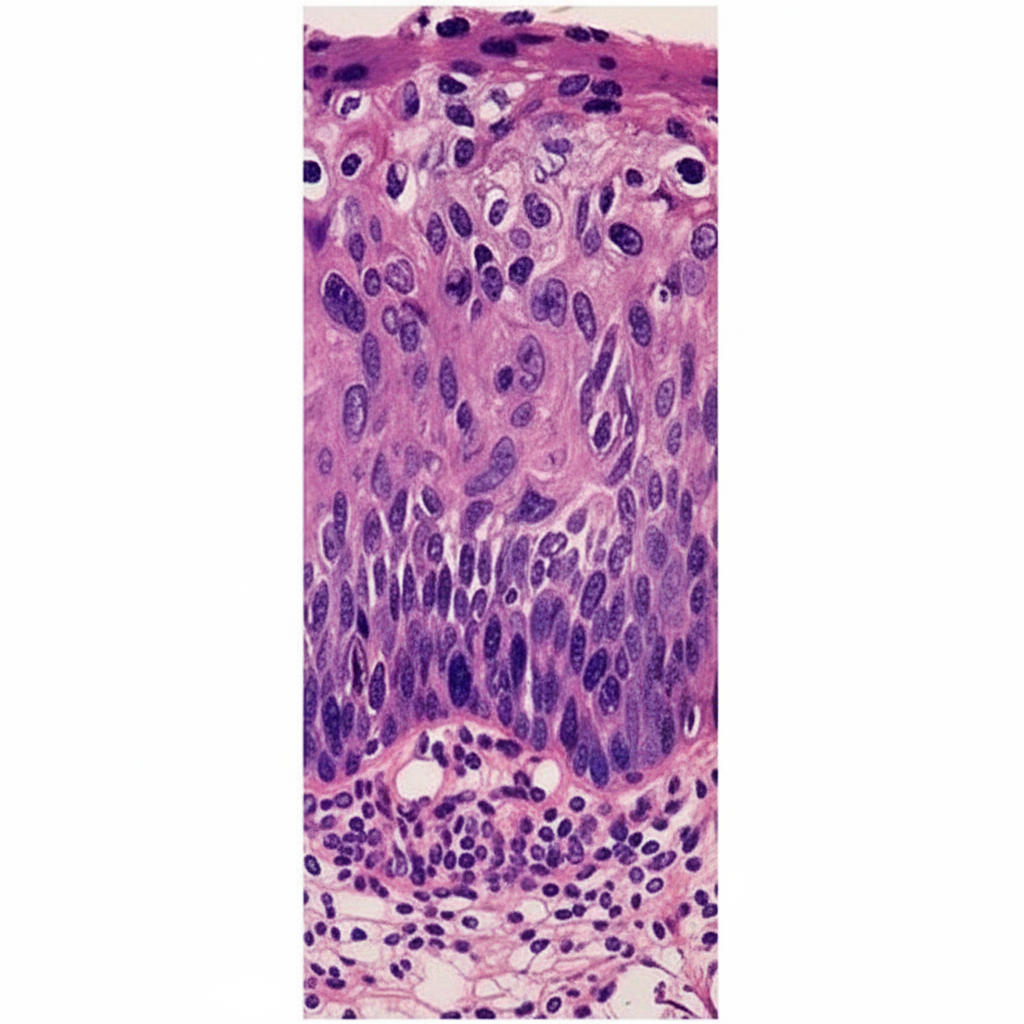
What is the earliest morphological evidence of ovulation?
Call-Exner bodies are seen in which of the following tumors?
Which testicular tumor is associated with high HCG levels?
Women carrying the BRCA1 gene are more likely to develop which type of breast carcinoma?
Streak gonads are seen in which of the following conditions?
Which of the following predisposes to breast cancer?
Practice by Chapter
Diseases of Male Genital Tract
Practice Questions
Testicular Tumors
Practice Questions
Prostate Pathology
Practice Questions
Diseases of Female Genital Tract
Practice Questions
Cervical Pathology and Neoplasia
Practice Questions
Endometrial Pathology
Practice Questions
Ovarian Diseases and Tumors
Practice Questions
Gestational Trophoblastic Disease
Practice Questions
Placental Pathology
Practice Questions
Sexually Transmitted Infections
Practice Questions
Want unlimited practice?
Get full access to all questions, explanations, and performance tracking.
Scan to download app
