Reproductive Pathology — MCQs

On this page
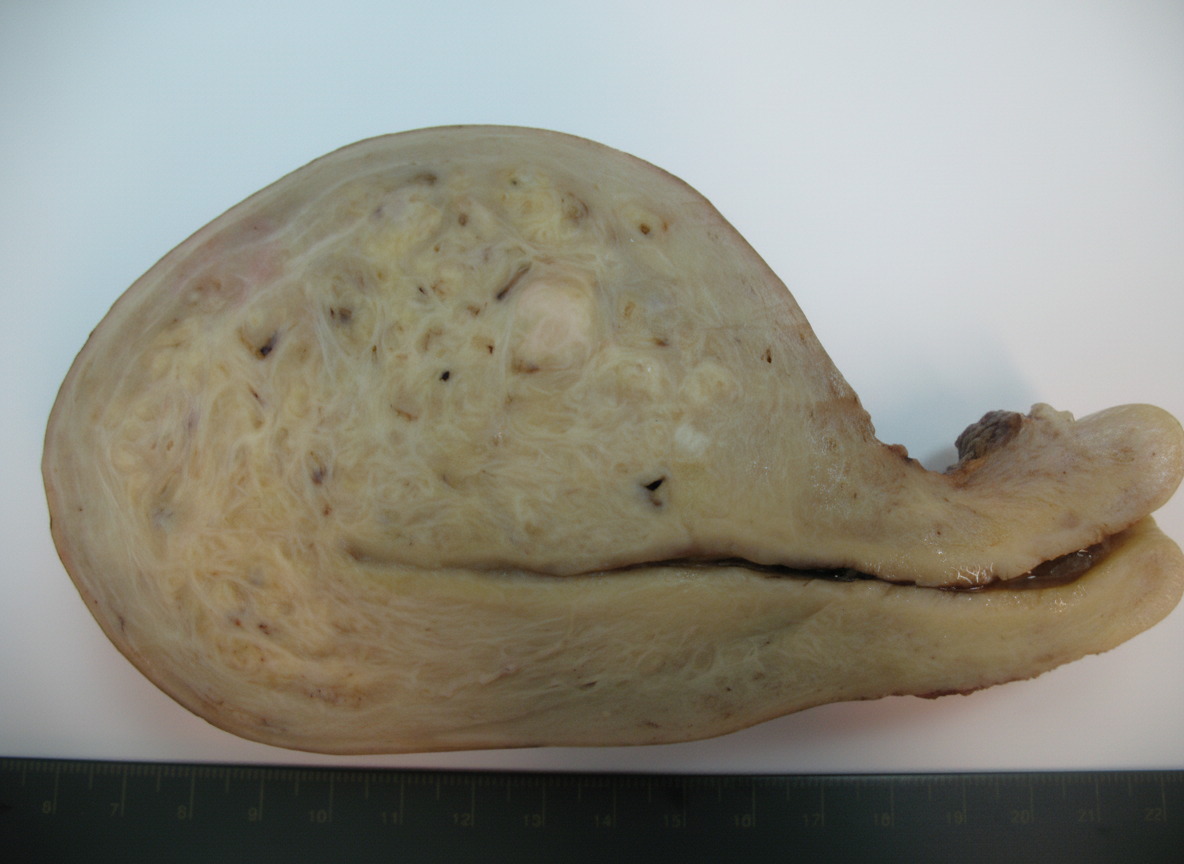
A hysterectomy specimen from a 38-year-old woman shows the following gross appearance. What is the most likely diagnosis?
Which of the following is not a sex cord-stromal tumor?
According to the Bethesda System, in 'LSIL' histologic changes are observed in which part of the cervix?
Biopsy from a testicular mass following orchidectomy showed sheets of uniform cells containing abundant clear cytoplasm with prominent nucleus. Which is the histopathological diagnosis?
What is the origin of cancer cells in a Krukenberg tumour?
What is true about the histology of infiltrating lobular breast carcinoma?
What condition presents the maximum risk of invasive breast carcinoma?
What is the most common age group for leiomyosarcoma?
Gleason's classification is used for which of the following conditions?
Which among the following are testicular tumor markers?
Practice by Chapter
Diseases of Male Genital Tract
Practice Questions
Testicular Tumors
Practice Questions
Prostate Pathology
Practice Questions
Diseases of Female Genital Tract
Practice Questions
Cervical Pathology and Neoplasia
Practice Questions
Endometrial Pathology
Practice Questions
Ovarian Diseases and Tumors
Practice Questions
Gestational Trophoblastic Disease
Practice Questions
Placental Pathology
Practice Questions
Sexually Transmitted Infections
Practice Questions
Want unlimited practice?
Get full access to all questions, explanations, and performance tracking.
Scan to download app
