Pancreatic Transplantation Pathology — MCQs

Hyperacute rejection occurs within:-
Most common complication after intestinal transplantation is
Most common infection post solid organ transplantation
Which of the following is not a recognized complication of chronic pancreatitis?
A 40-year-old man underwent kidney transplantation. Two months after transplantation, he developed fever and features suggestive of bilateral diffuse interstitial pneumonia. Which of the following is the most likely etiologic agent?
A chronic alcoholic patient came to emergency with severe pain in epigastrium and multiple episodes of vomiting. On examination, guarding was present in upper epigastrium. Chest X-ray was normal. What is the next best step?
Which is the cell of origin of Chronic Lymphocytic Leukaemia / Small Lymphocytic Lymphoma?
Glanzmann thrombasthenia is due to defect in:-
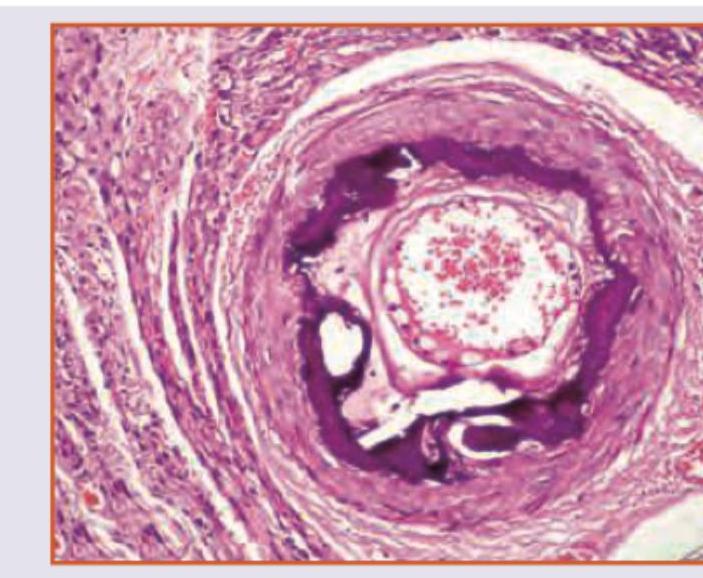
Which is true about the image shown?
Intraductal papillary mucinous neoplasm is a precursor of which of the following entities?
Want unlimited practice?
Get full access to all questions, explanations, and performance tracking.
Scan to download app
