Chronic Pancreatitis — MCQs

A 55-year-old white woman has had recurrent episodes of alcohol-induced pancreatitis. Despite abstinence, the patient develops postprandial abdominal pain, bloating, weight loss despite good appetite, and bulky, foul-smelling stools. Kidney, ureter, bladder (KUB) x-ray shows pancreatic calcifications. In this patient, you should expect to find which of the following?
Cells central to the production of pancreatic fibrosis are?
Pancreatitis is a common complication of which one of the following?
Which of the following is not a recognized complication of chronic pancreatitis?
Which is not a component of Ranson's criteria for acute pancreatitis?
A 55-year-old man presents with intermittent epigastric pain, relieved by eating, and worsened by fasting. What is the most likely cause?
A chronic alcoholic patient came to emergency with severe pain in epigastrium and multiple episodes of vomiting. On examination, guarding was present in upper epigastrium. Chest X-ray was normal. What is the next best step?
In cystic fibrosis, which of the following structures is affected in the pancreas?
Type of necrosis in pancreatitis-
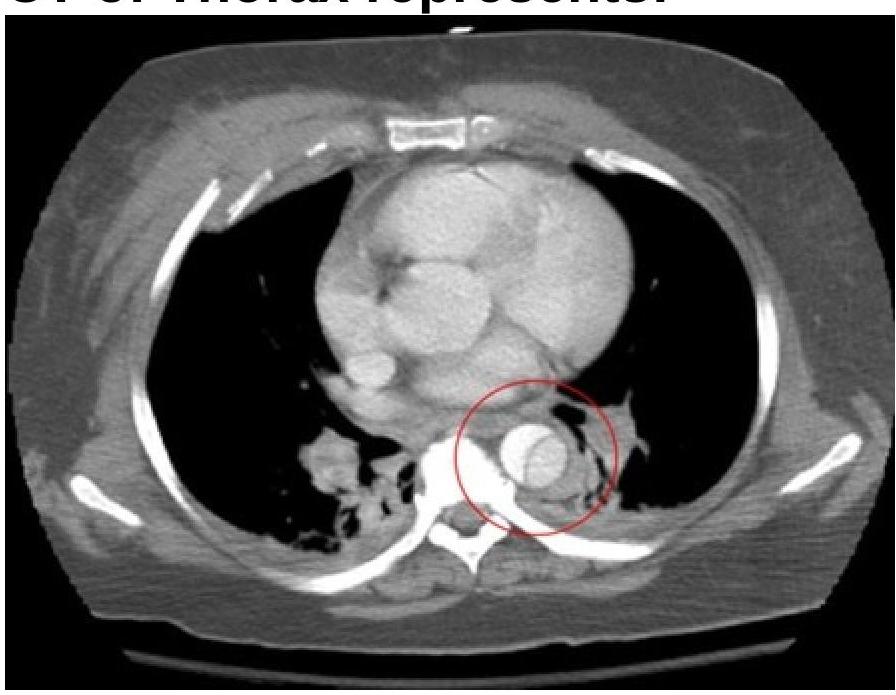
The CT thorax image shows:
Want unlimited practice?
Get full access to all questions, explanations, and performance tracking.
Scan to download app
