Molecular Pathology — MCQs

On this page
A 22-year-old woman presents with congenital anemia requiring multiple RBC transfusions for many years. On physical examination, her skin has a bronze color and liver function tests show reduced serum albumin. Which of the following findings would most likely appear in a liver biopsy specimen?
Mutation in the gene encoding Dystrophin is known to cause all the following except?
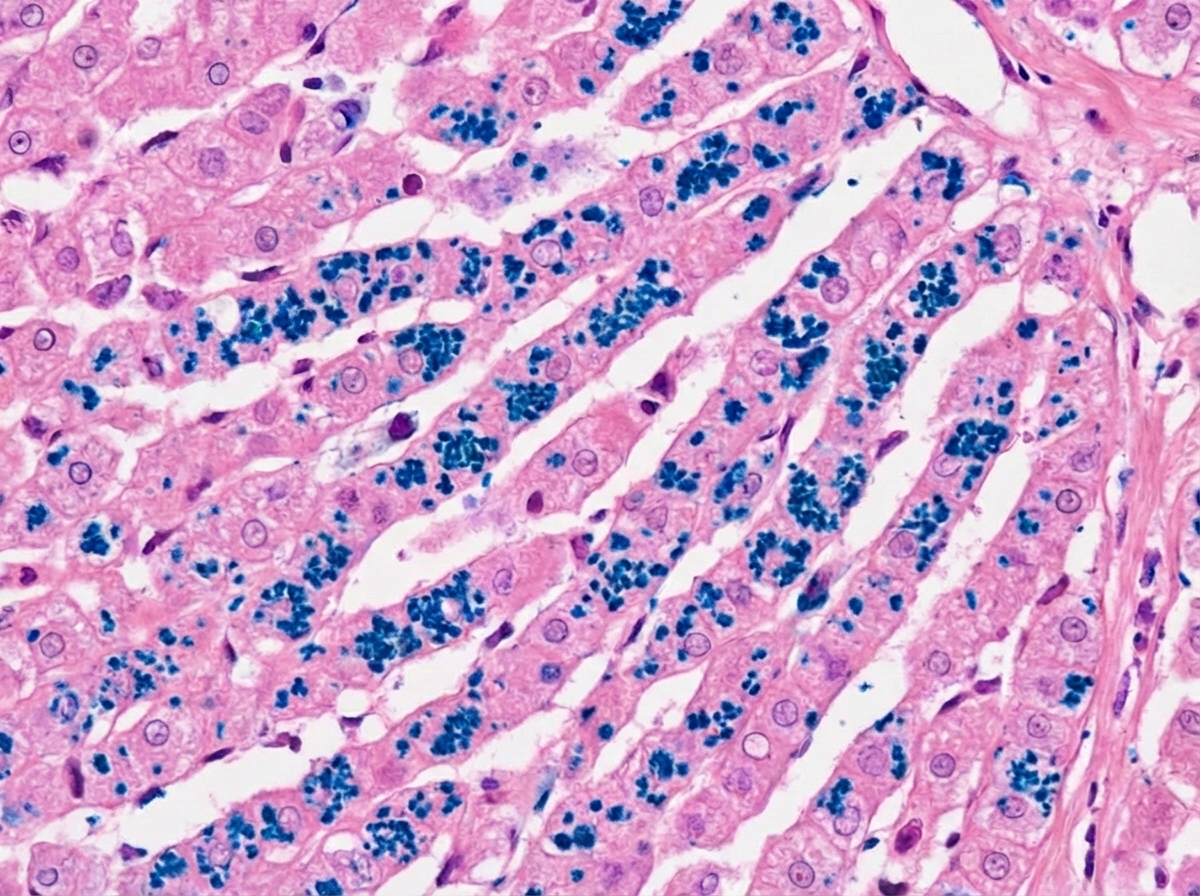
Given below is the histopathology of liver biopsy of hemochromatosis. Which of the following stain is used to detect iron deposition?
Councilman bodies are formed due to which process?
Liquefactive necrosis is seen in which of the following organs?
Which is the most common chromosomal anomaly seen?
All of the following are methods of cellular adaptation EXCEPT?
Which of the following can be used as an immunohistochemical marker for juvenile rhabdomyosarcoma?
Annexin V is a marker of?
Jumping genes (transposons) are primarily involved in which of the following biological processes?
Practice by Chapter
Principles of Molecular Pathology
Practice Questions
DNA and RNA Analysis Techniques
Practice Questions
Cytogenetics
Practice Questions
Polymerase Chain Reaction Applications
Practice Questions
Next-Generation Sequencing
Practice Questions
Molecular Diagnosis of Infectious Diseases
Practice Questions
Molecular Oncology
Practice Questions
Pharmacogenomics
Practice Questions
Genetic Counseling and Risk Assessment
Practice Questions
Molecular Diagnostics Quality Control
Practice Questions
Want unlimited practice?
Get full access to all questions, explanations, and performance tracking.
Scan to download app
