Molecular Pathology — MCQs

On this page
Gene instability associated with malignancy is seen in which of the following conditions?
What is the target gene that is affected by the E6 gene product of HPV, leading to malignant transformation?
Best sample for karyotyping is?
Which of the following methods is commonly used to study oncogenes?
Which of the following genes is least likely to be involved in the development of carcinoma of the colon?
Which genetic mutation is most commonly associated with male breast carcinoma?
The gene most commonly involved in endometrial carcinoma is:
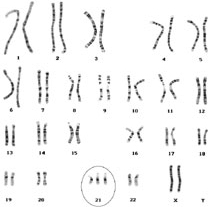
The given karyotype is seen in which of the following syndromes?
In breast cancer, Her-2/Neu promotes tumorigenesis by what mechanism?
Which of the following is not a chromosome breakage disorder?
Practice by Chapter
Principles of Molecular Pathology
Practice Questions
DNA and RNA Analysis Techniques
Practice Questions
Cytogenetics
Practice Questions
Polymerase Chain Reaction Applications
Practice Questions
Next-Generation Sequencing
Practice Questions
Molecular Diagnosis of Infectious Diseases
Practice Questions
Molecular Oncology
Practice Questions
Pharmacogenomics
Practice Questions
Genetic Counseling and Risk Assessment
Practice Questions
Molecular Diagnostics Quality Control
Practice Questions
Want unlimited practice?
Get full access to all questions, explanations, and performance tracking.
Scan to download app
