Molecular Pathology — MCQs

On this page
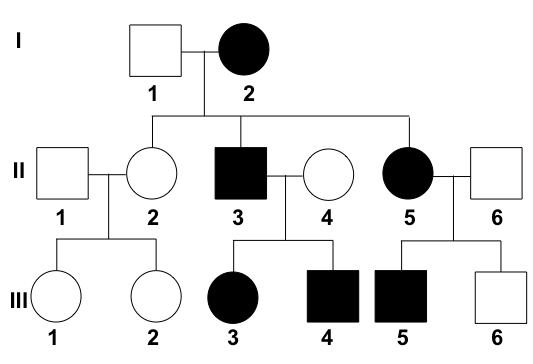
Analyze the provided pedigree chart to identify the underlying genetic disease.
What is the most common tumor in females with tuberous sclerosis?
What is a Leiden mutation?
All of the following are intermediate filaments except?
What is the gene involved in GIST?
A patient died of Alzheimer's disease. At autopsy, the heart contains a yellow-brown, finely granular pigment. What is the most likely cause of this pigment?
Which of the following is a sign of chronic inflammation?
What is the function of the FMR1 protein?
Ring sideroblasts in myelodysplastic syndrome are associated with mutations in which gene?
Which stain is used for tissue fats?
Practice by Chapter
Principles of Molecular Pathology
Practice Questions
DNA and RNA Analysis Techniques
Practice Questions
Cytogenetics
Practice Questions
Polymerase Chain Reaction Applications
Practice Questions
Next-Generation Sequencing
Practice Questions
Molecular Diagnosis of Infectious Diseases
Practice Questions
Molecular Oncology
Practice Questions
Pharmacogenomics
Practice Questions
Genetic Counseling and Risk Assessment
Practice Questions
Molecular Diagnostics Quality Control
Practice Questions
Want unlimited practice?
Get full access to all questions, explanations, and performance tracking.
Scan to download app
