Inflammation and Repair — MCQs

On this page
A 29-year-old carpenter receives a traumatic laceration to her left arm. Which of the following is the most important factor that determines whether this wound will heal by primary or secondary intention?
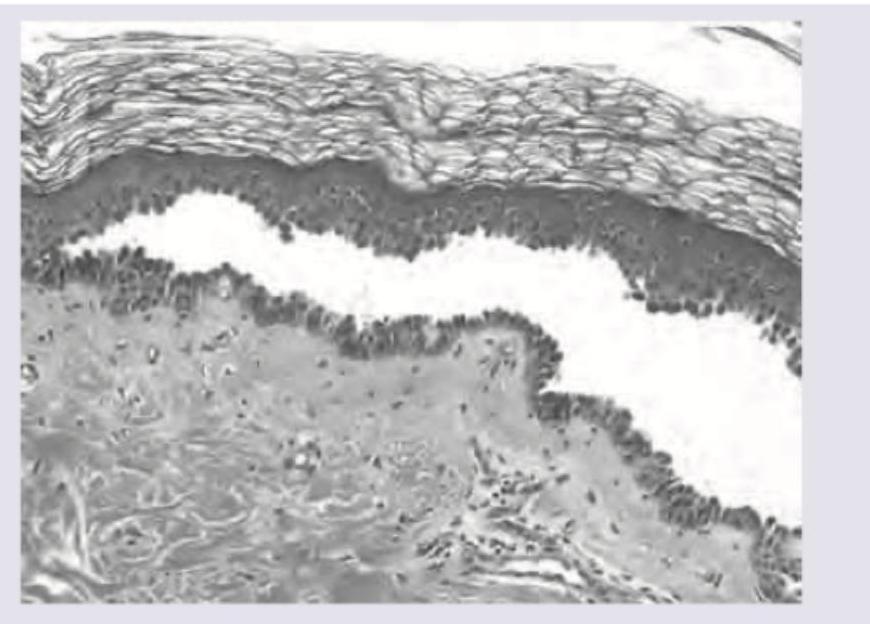
The morphological pattern of inflammation shown here is:
Provisional matrix is made up of which of the following components?
What is the primary type of collagen found in early granulation tissue?
Which of the following statements regarding keloid formation is true?
Which of the following is NOT seen in the inflammatory stage of wound healing?
Which of the following statements regarding primary intentional healing is true?
In healing by first intention, all of the following occur EXCEPT?
Which of the following is NOT an action of TGF-beta?
Which of the following chemical mediators of inflammation is an example of a C-X-C or alpha chemokine?
Practice by Chapter
Acute Inflammation: Vascular Events
Practice Questions
Acute Inflammation: Cellular Events
Practice Questions
Chemical Mediators of Inflammation
Practice Questions
Chronic Inflammation
Practice Questions
Granulomatous Inflammation
Practice Questions
Systemic Effects of Inflammation
Practice Questions
Wound Healing
Practice Questions
Tissue Regeneration
Practice Questions
Fibrosis and Repair
Practice Questions
Resolution of Inflammation
Practice Questions
Want unlimited practice?
Get full access to all questions, explanations, and performance tracking.
Scan to download app
