Hematopathology — MCQs

On this page
Heterozygous sickle cell anemia provides protection against which of the following?
Which subtype of Acute Myeloid Leukemia (AML) is characterized by gum infiltration and hepatosplenomegaly?
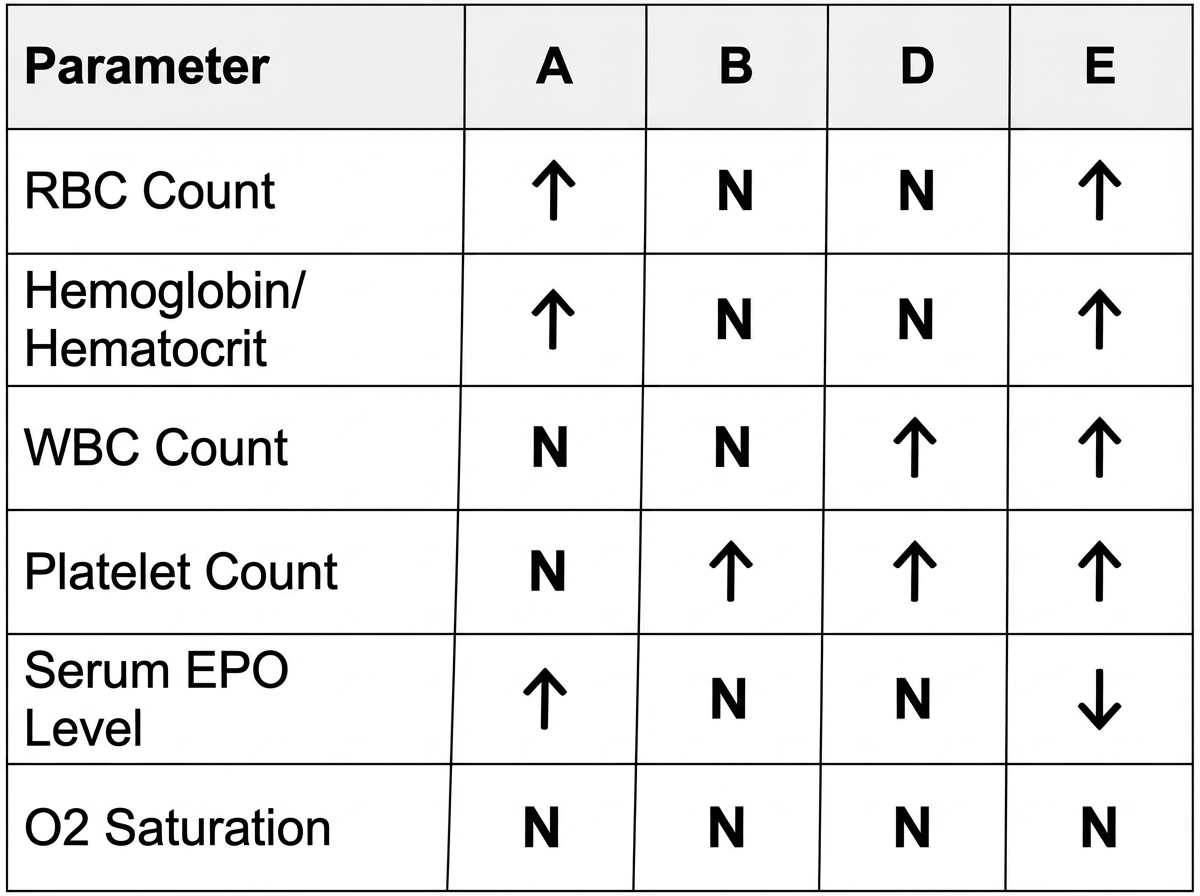
Which one of the labeled boxes in the diagram below is most consistent with the expected findings for an individual with polycythemia rubra vera?
Defect in Bernard Soulier syndrome lies in which of the following?
Which of the following conditions is characterized by an intrinsic cell wall defect of red blood cells?
The cell mass in multiple myeloma is formed by?
Nuclear-cytoplasmic asynchrony is usually seen in:
Bone marrow showing positive staining with Periodic Acid Schiff. What is the likely diagnosis?
Dohle bodies are seen in which of the following conditions?
Acquired mutations in the PIGA gene give rise to which condition?
Practice by Chapter
Anemias: Classification and Approach
Practice Questions
Hemolytic Anemias
Practice Questions
Myeloproliferative Neoplasms
Practice Questions
Myelodysplastic Syndromes
Practice Questions
Acute Leukemias
Practice Questions
Chronic Leukemias
Practice Questions
Lymphomas and Lymphoid Neoplasms
Practice Questions
Plasma Cell Disorders
Practice Questions
Bleeding Disorders
Practice Questions
Thrombotic Disorders
Practice Questions
Want unlimited practice?
Get full access to all questions, explanations, and performance tracking.
Scan to download app
