Hematopathology — MCQs

On this page
Spot the diagnosis?
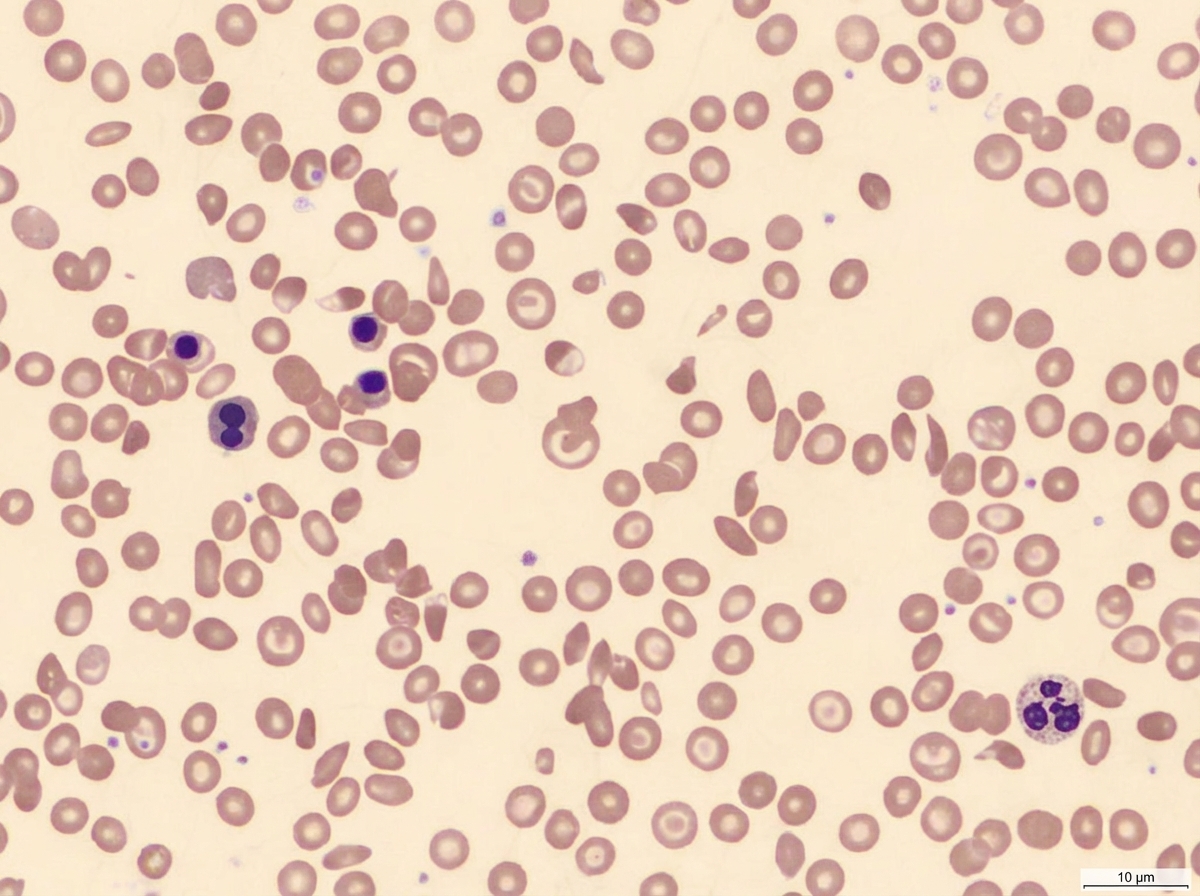
Spur cells are seen in which of the following conditions?
A 48-year-old woman complains of weakness, fatigue, and easy bruisability for 2 months. She had worked as a technician in a nuclear energy plant for 15 years and was involved in an accident during which she was exposed to considerable radiation. Physical examination reveals an enlarged liver and spleen. What disease is the likely cause of her condition?
What is the reason for hydrops fetalis in Hb Barts disease?
A patient presents with Hb level 6 g/dL, TLC 3500/mm 3, and platelet count 50,000/mm 3. What is the most probable diagnosis?
A 50-year-old man presents with fever and diffuse lymphadenopathy. A lymph node biopsy reveals non-Hodgkin follicular lymphoma. Immunohistochemical staining of neoplastic lymphoid cells within the nodular areas of the lymph node would be expected to stain positively for which of the following protein markers?
HbE is common in which region of India?
The most striking haematological finding in agranulocytosis is?
Which syndrome is not associated with leukemia?
What is the best screening test for hemophilia?
Practice by Chapter
Anemias: Classification and Approach
Practice Questions
Hemolytic Anemias
Practice Questions
Myeloproliferative Neoplasms
Practice Questions
Myelodysplastic Syndromes
Practice Questions
Acute Leukemias
Practice Questions
Chronic Leukemias
Practice Questions
Lymphomas and Lymphoid Neoplasms
Practice Questions
Plasma Cell Disorders
Practice Questions
Bleeding Disorders
Practice Questions
Thrombotic Disorders
Practice Questions
Want unlimited practice?
Get full access to all questions, explanations, and performance tracking.
Scan to download app
