Hematopathology — MCQs

On this page
This is the peripheral smear of which disease?
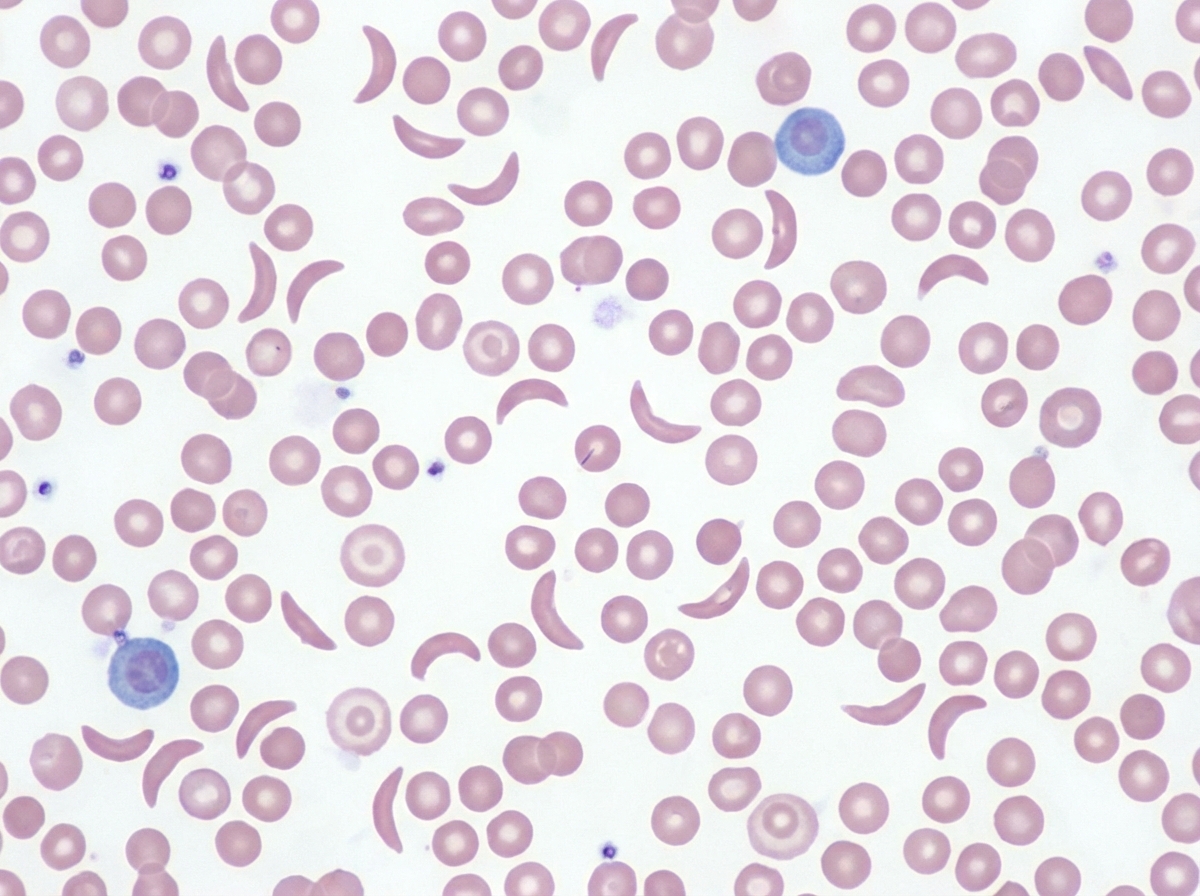
BCR-ABL1 gene fusion is seen in all of the following conditions except?
Which type of Hodgkin's disease is commonest in females and associated with an excellent prognosis?
Which coagulation test is typically prolonged in hemophilia?
Which subtype of Hodgkin lymphoma is associated with the maximum incidence of 'B' symptoms?
Bence Jones proteinuria may be seen in:
Which of the following is NOT a characteristic feature of multiple myeloma?
Splenic auto-infarction is typically seen in which of the following conditions?
Raised aPTT is seen in all of the following bleeding disorders except?
What is the highest level of LAP seen in?
Practice by Chapter
Anemias: Classification and Approach
Practice Questions
Hemolytic Anemias
Practice Questions
Myeloproliferative Neoplasms
Practice Questions
Myelodysplastic Syndromes
Practice Questions
Acute Leukemias
Practice Questions
Chronic Leukemias
Practice Questions
Lymphomas and Lymphoid Neoplasms
Practice Questions
Plasma Cell Disorders
Practice Questions
Bleeding Disorders
Practice Questions
Thrombotic Disorders
Practice Questions
Want unlimited practice?
Get full access to all questions, explanations, and performance tracking.
Scan to download app
