Hematopathology — MCQs

On this page
Which of the following features is shared in common between lymphocyte-rich and lymphocyte-predominant types of Hodgkin's lymphoma?
All of the following statements about the Philadelphia chromosome in CML are true, except:
Working formulation in staging of non-Hodgkin's lymphoma is based on which of the following?
What is true about follicular lymphoma?
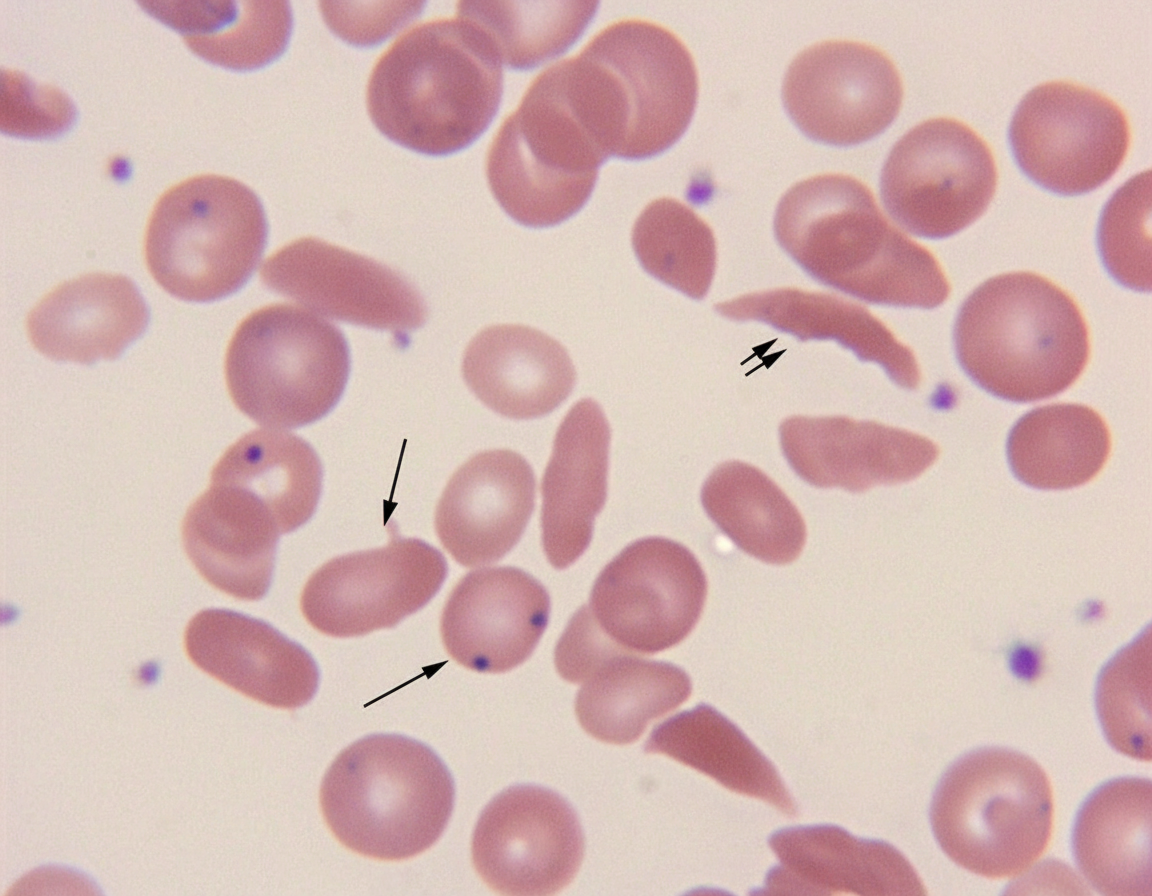
The above marked structures are seen in all except?
Non-specific esterase is positive in all categories of AML except?
Popcorn cells are characteristic of which of the following hematological malignancies?
Reticulocytes are stained by which method?
Radiological examination shows evidence of bone infarct in a child. Which of the following conditions may be responsible?
Which of the following is NOT typically seen in Disseminated Intravascular Coagulation (DIC)?
Practice by Chapter
Anemias: Classification and Approach
Practice Questions
Hemolytic Anemias
Practice Questions
Myeloproliferative Neoplasms
Practice Questions
Myelodysplastic Syndromes
Practice Questions
Acute Leukemias
Practice Questions
Chronic Leukemias
Practice Questions
Lymphomas and Lymphoid Neoplasms
Practice Questions
Plasma Cell Disorders
Practice Questions
Bleeding Disorders
Practice Questions
Thrombotic Disorders
Practice Questions
Want unlimited practice?
Get full access to all questions, explanations, and performance tracking.
Scan to download app
