Hematopathology — MCQs

On this page
CD19 positive, CD22 positive, CD103 positive monoclonal B-cells with bright kappa positivity were found to comprise 60% of the peripheral blood lymphoid cells on flow cytometric analysis in a 55-year-old man with massive splenomegaly and a total leucocyte count of 3.3 x 10^9/L. Which one of the following is the most likely diagnosis?
Which of the following statements about stored blood is false?
ADAMTS13 is associated with which of the following disorders?
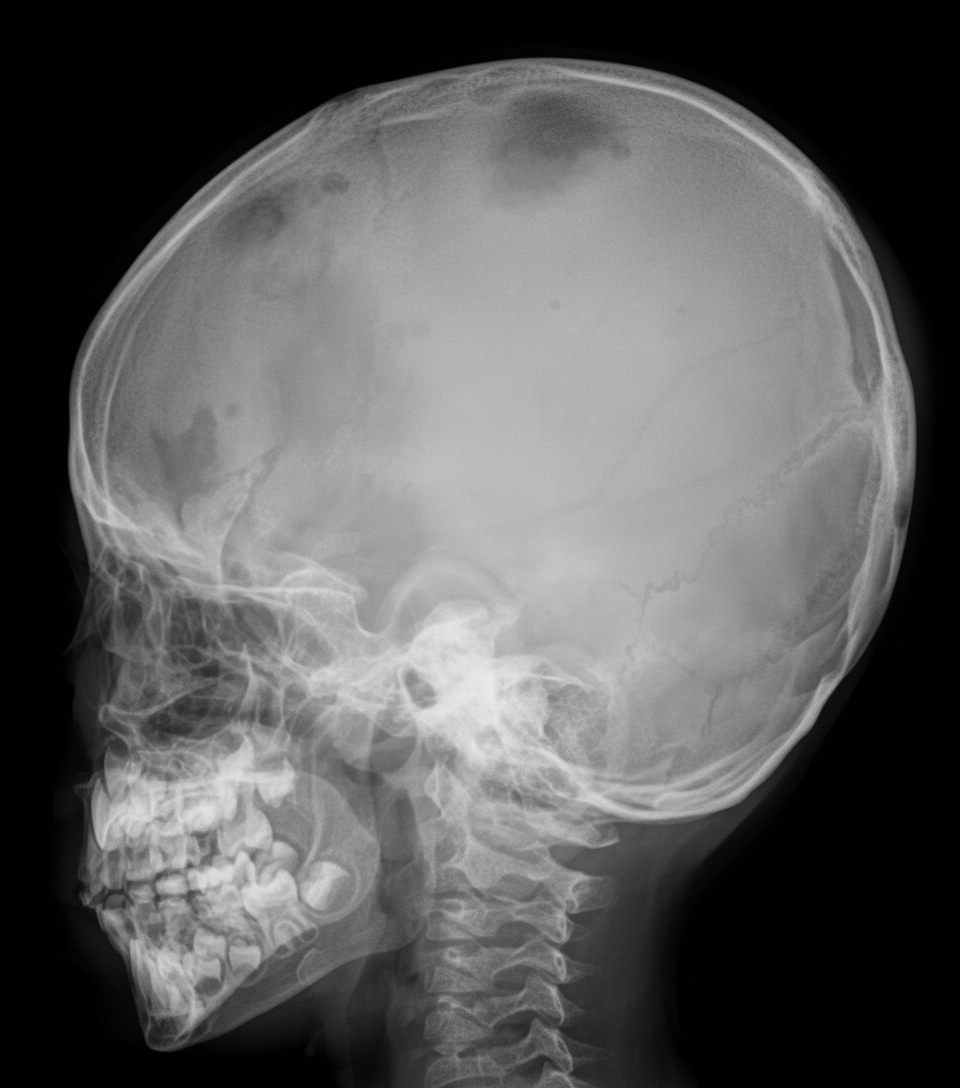
An infant presents with bony pain and dermatitis on the scalp. Skull X-ray and electron microscopy of a biopsy from a lesion are provided. What is the most probable diagnosis?
Which of the following is NOT a cause of Disseminated Intravascular Coagulation (DIC)?
All are seen in Thalassemia major EXCEPT?
"Autosplenectomy" is a feature seen in:
What is the characteristic finding in chronic myeloid leukemia?
Auer rods are specific for which of the following conditions?
Plasmacytoid lymphomas may be associated with which immunoglobulin?
Practice by Chapter
Anemias: Classification and Approach
Practice Questions
Hemolytic Anemias
Practice Questions
Myeloproliferative Neoplasms
Practice Questions
Myelodysplastic Syndromes
Practice Questions
Acute Leukemias
Practice Questions
Chronic Leukemias
Practice Questions
Lymphomas and Lymphoid Neoplasms
Practice Questions
Plasma Cell Disorders
Practice Questions
Bleeding Disorders
Practice Questions
Thrombotic Disorders
Practice Questions
Want unlimited practice?
Get full access to all questions, explanations, and performance tracking.
Scan to download app
