Hematopathology — MCQs

On this page
In a patient with general fatigue, normal TLC/ DLC, and superficial discrete lymphadenopathy, with lymph node biopsy showing effaced architecture, atypical cells with indented nuclei and prominent nucleoli, positive for CD10 and BCL-2, which of the following is the most likely diagnosis?
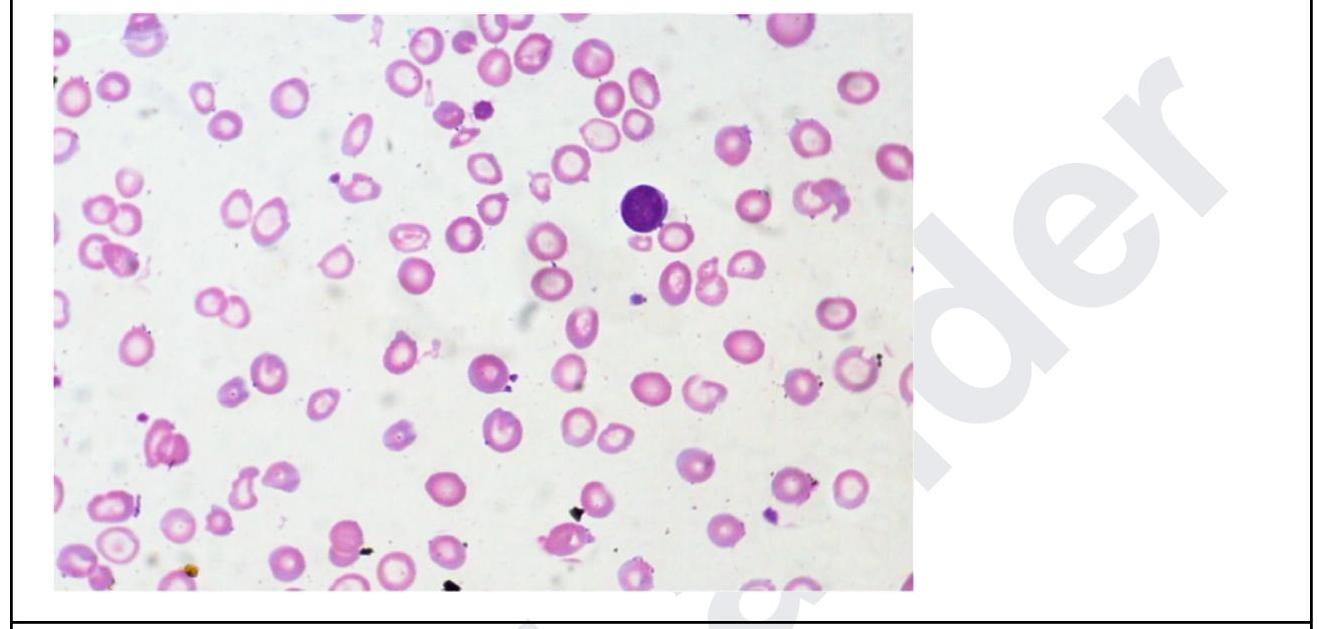
A 35 year old woman presents with fatigue. Investigations revealed the following: Hb, 5 g/dL; MCH, 24; low MCV; leukocytes, 11,000/ uL, and platelets, 5 lakhs. The peripheral smear is shown below. What is the diagnosis? Normal values: - Mean cell volume (MCV); 90 ± 8 fL - Mean cell Hb(MCH); 30 ± 3 pg
Match the following cell types/patterns (Column A) with their associated malignancies (Column B): Column A (Cell types/patterns): a) Faggot cell b) Popcorn cell c) Starry sky pattern d) Cerebriform nuclei Column B (Associated malignancies): 1) Acute promyelocytic leukemia 2) Lymphocyte-predominant Hodgkin's lymphoma 3) Burkitt lymphoma 4) Sezary syndrome
The Hb is 5 g/dL and the reticulocyte count is 9%. What is the corrected reticulocyte count?
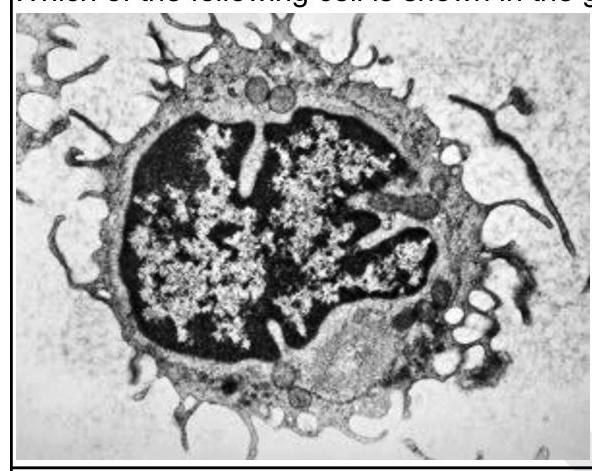
Which of the following cells is shown in the given image?
Which of the following are characteristic laboratory findings in Iron Deficiency Anemia (IDA)? 1. Low serum ferritin 2. Low transferrin saturation 3. Low serum iron 4. Increased TIBC
The graphical representation for flow cytometry analysis is done by which of the following?
A boy presents with fever, night sweats, and neck swelling. The biopsy of swelling showed a starry sky appearance. What is the most likely genetic abnormality seen in this case?
A patient with lytic lesions on the skull is suspected of a diagnosis of Langerhans cell histiocytosis. Which of the following is a characteristic finding on electron microscopy?
Which is the cell of origin of Chronic Lymphocytic Leukaemia / Small Lymphocytic Lymphoma?
Practice by Chapter
Anemias: Classification and Approach
Practice Questions
Hemolytic Anemias
Practice Questions
Myeloproliferative Neoplasms
Practice Questions
Myelodysplastic Syndromes
Practice Questions
Acute Leukemias
Practice Questions
Chronic Leukemias
Practice Questions
Lymphomas and Lymphoid Neoplasms
Practice Questions
Plasma Cell Disorders
Practice Questions
Bleeding Disorders
Practice Questions
Thrombotic Disorders
Practice Questions
Want unlimited practice?
Get full access to all questions, explanations, and performance tracking.
Scan to download app
