Hematopathology — MCQs

On this page
Which of the following is the most common chromosomal abnormality associated with acute myeloid leukemia (AML) with a favorable prognosis?
A 68-year-old man presents with bleeding manifestations. Peripheral smear shows the presence of cells shown below. Which of the following is the most common chromosomal abnormality seen in this condition?
Which of the following laboratory techniques is most commonly used to compare and quantify CD markers on cells?
A lymphoma characterized by the presence of centrocytes and centroblasts, along with BCL2 positivity and CD10 expression, is most commonly associated with which chromosomal translocation?
Which of the following hematological findings are seen in pregnant women with thalassemia trait?
Folic acid deficiency is characterized by the following features except
Basophilic stippling of the RBCs is a sensitive index of
Which of the following blood components has the shortest shelf life?
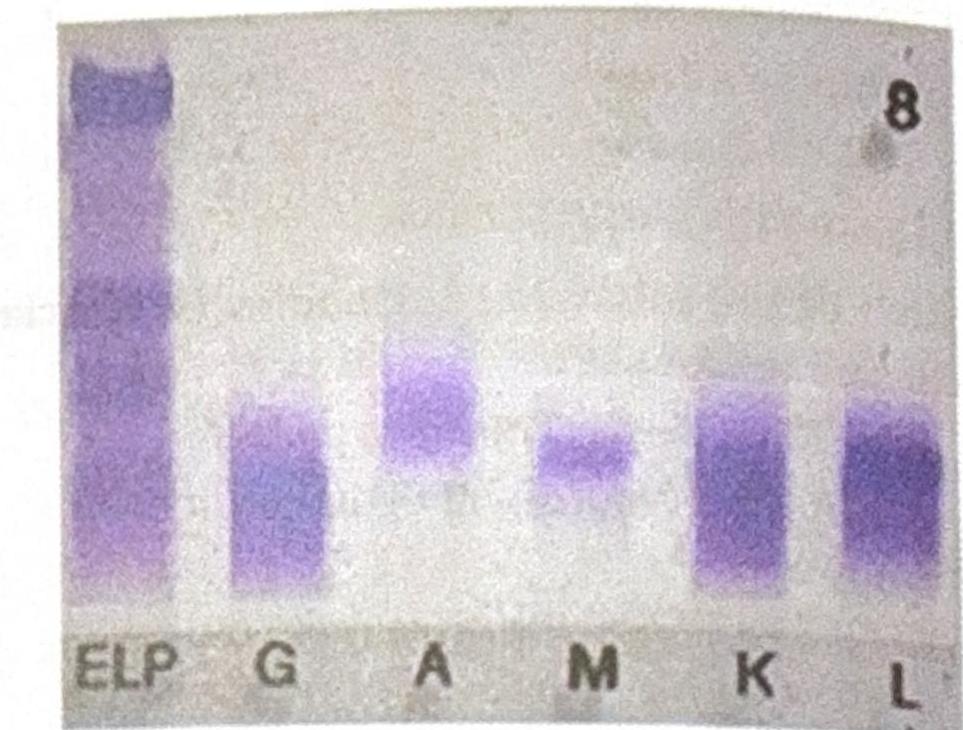
An elderly male patient presented with clinical symptoms and signs consistent with possible multiple myeloma. Electrophoresis shows an M spike, and immunofixation findings are shown below. Which of the following statements best corresponds to the findings?
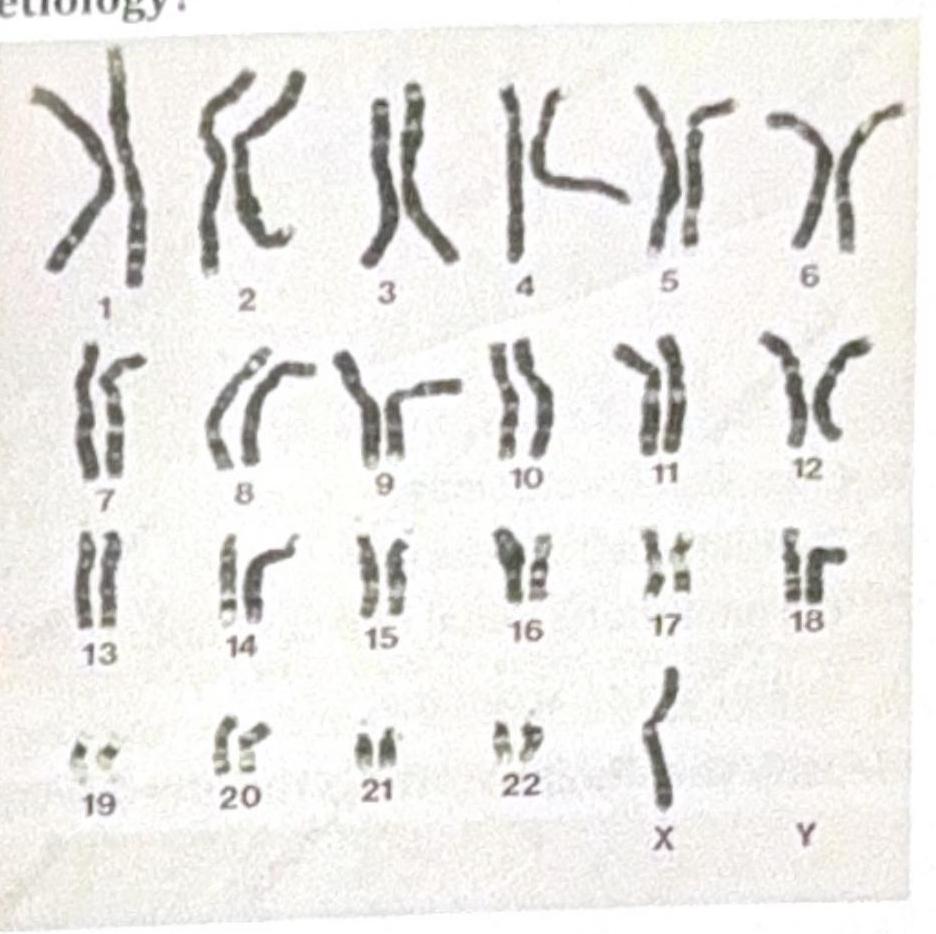
Identify the image and the disease it is associated with: 
Practice by Chapter
Anemias: Classification and Approach
Practice Questions
Hemolytic Anemias
Practice Questions
Myeloproliferative Neoplasms
Practice Questions
Myelodysplastic Syndromes
Practice Questions
Acute Leukemias
Practice Questions
Chronic Leukemias
Practice Questions
Lymphomas and Lymphoid Neoplasms
Practice Questions
Plasma Cell Disorders
Practice Questions
Bleeding Disorders
Practice Questions
Thrombotic Disorders
Practice Questions
Want unlimited practice?
Get full access to all questions, explanations, and performance tracking.
Scan to download app
