Hematopathology — MCQs

On this page
Disseminated Intravascular Coagulation (DIC) is seen with all of the following conditions EXCEPT:
Which anticoagulant is used for chelating calcium?
Which of the following conditions predisposes to leukemia?
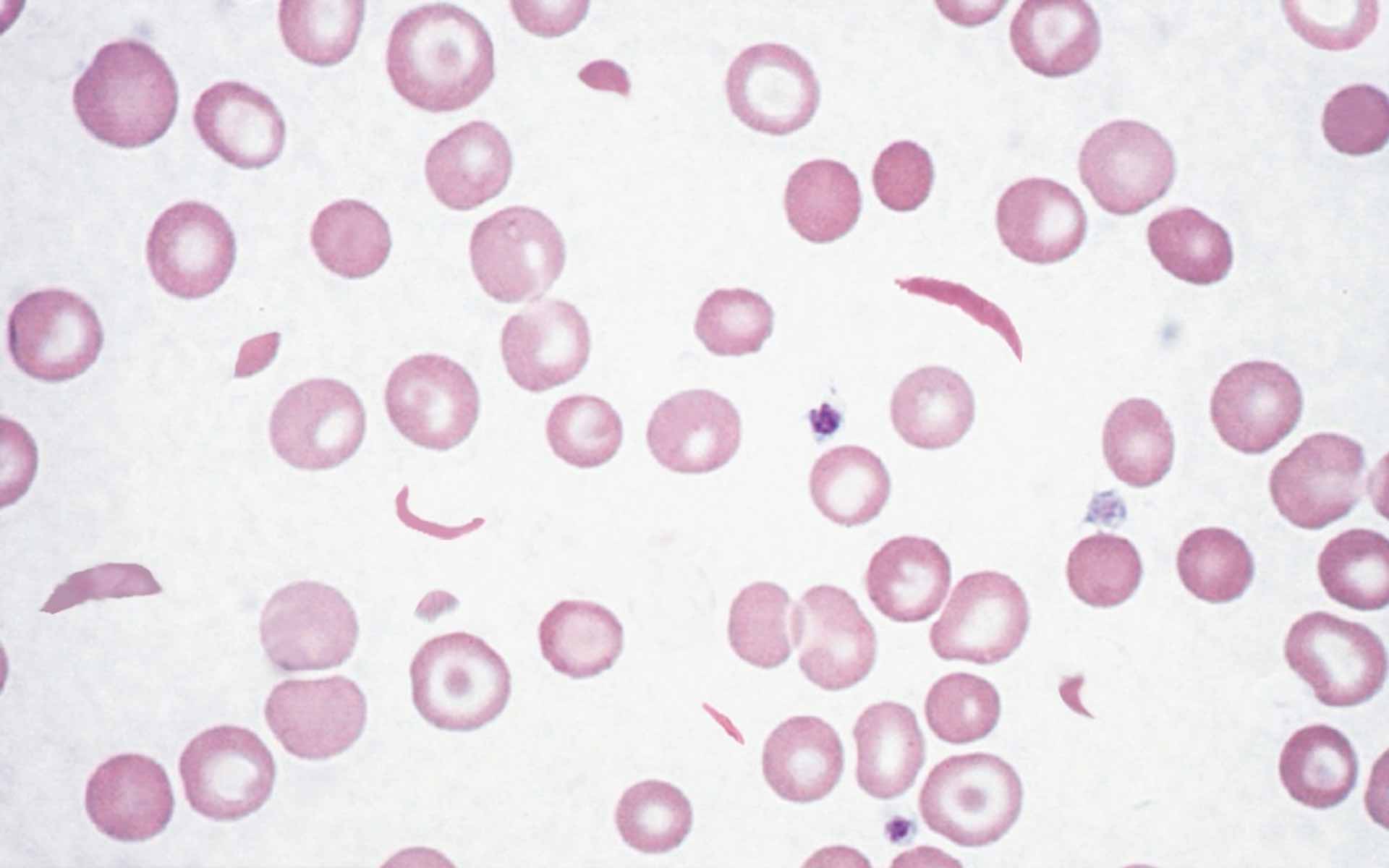
Which organ is most commonly affected in the disorder characterized by the given peripheral blood smear findings?
In leucopenia, which white blood cell type is predominantly involved?
Which cell is not seen in Hodgkin lymphoma?
Lupus anticoagulants may cause all of the following except:
Reticulocytes are stained with which of the following reagents?
Which of the viruses is not commonly implicated in the pathogenesis of Non-Hodgkin's lymphomas?
Histiocytosis X is a spectrum of disorders that includes which of the following conditions?
Practice by Chapter
Anemias: Classification and Approach
Practice Questions
Hemolytic Anemias
Practice Questions
Myeloproliferative Neoplasms
Practice Questions
Myelodysplastic Syndromes
Practice Questions
Acute Leukemias
Practice Questions
Chronic Leukemias
Practice Questions
Lymphomas and Lymphoid Neoplasms
Practice Questions
Plasma Cell Disorders
Practice Questions
Bleeding Disorders
Practice Questions
Thrombotic Disorders
Practice Questions
Want unlimited practice?
Get full access to all questions, explanations, and performance tracking.
Scan to download app
