Hematopathology — MCQs

On this page
Target cells are seen in which of the following conditions?
Bence Jones proteinuria is found on a urinalysis specimen from a 63-year-old woman who is developing chronic renal failure with high serum urea nitrogen and creatinine. A radiograph of the vertebral column demonstrates multiple 1 to 2 cm rounded areas of decreased bone density. The pathological examination from a renal biopsy is likely to show which of the following complications?
In sickle cell anaemia, the defect is in which globin chain?
A patient presents with a lytic bone lesion. Histology reveals monoclonal plasma cells. Bone marrow biopsy shows less than 10 percent plasma cells, and a small M spike is reported on electrophoresis. Skeletal survey is otherwise unremarkable. What is the most likely diagnosis?
Platelets can be stored at what temperature and for how long?
At what concentration of methemoglobin does cyanosis appear?
A 50-year-old male presented with left upper quadrant pain. On examination, the spleen was palpable 10 cm below the left costal margin. A peripheral smear is shown. Which of the following immunohistochemical markers is used for the diagnosis of the condition?
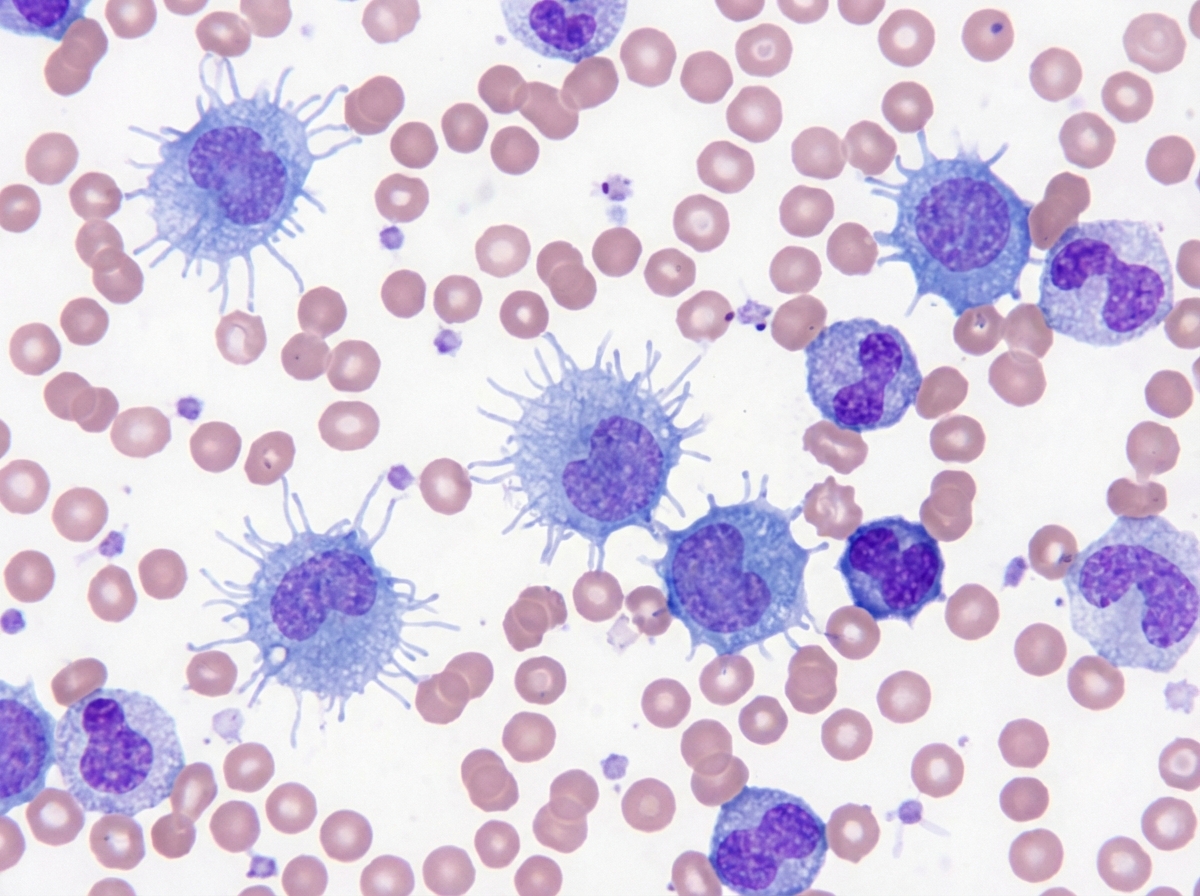
Paroxysmal nocturnal hemoglobinuria (PNH) is a disease that results from defects in which of the following?
A 10-year-old male child presents with pallor and a history of blood transfusion 2 months back. On investigation, Hb is 4.5 gms, total count is 60,000, and platelet count is 2 lakhs. Immunophenotyping shows CD10+, CD19+, CD117+, MPO+, and CD33-. What is the most probable diagnosis?
An 80-year-old asymptomatic woman on routine examination was detected to have a monoclonal spike on serum electrophoresis (IgG levels 1.5 g/dl). Bone marrow revealed 8% plasma cells. Which of the following represents the most likely diagnosis?
Practice by Chapter
Anemias: Classification and Approach
Practice Questions
Hemolytic Anemias
Practice Questions
Myeloproliferative Neoplasms
Practice Questions
Myelodysplastic Syndromes
Practice Questions
Acute Leukemias
Practice Questions
Chronic Leukemias
Practice Questions
Lymphomas and Lymphoid Neoplasms
Practice Questions
Plasma Cell Disorders
Practice Questions
Bleeding Disorders
Practice Questions
Thrombotic Disorders
Practice Questions
Want unlimited practice?
Get full access to all questions, explanations, and performance tracking.
Scan to download app
