Hematopathology — MCQs

On this page
CD-10 is seen in which of the following conditions?
Platelets are stored at what temperature?
Translocation of the bcr-abl gene is characteristically seen in which of the following conditions?
Burr cells are seen in which condition?
Gandy Gamma bodies are seen in which of the following conditions?
Disseminated intravascular coagulation (DIC) is commonly seen in which of the following conditions?
In megaloblastic anemia, what is the characteristic cell morphology?
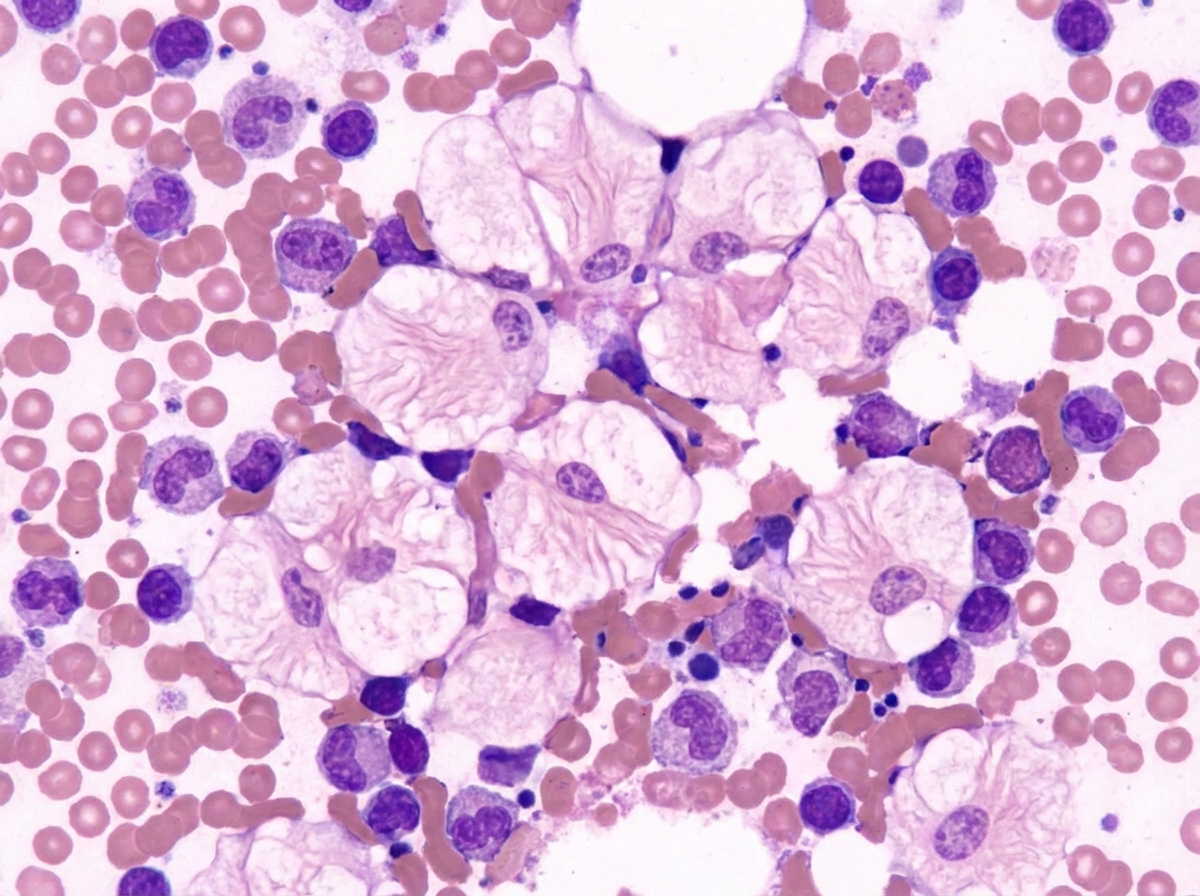
A 3-year-old child presented with anemia and thrombocytopenia. On examination, there was massive splenomegaly. A bone marrow aspiration revealed specific cellular findings. What is the diagnosis?
What is the investigation of choice to confirm sickle cell disease?
What type of antibodies are typically found in Idiopathic Thrombocytopenic Purpura (ITP)?
Practice by Chapter
Anemias: Classification and Approach
Practice Questions
Hemolytic Anemias
Practice Questions
Myeloproliferative Neoplasms
Practice Questions
Myelodysplastic Syndromes
Practice Questions
Acute Leukemias
Practice Questions
Chronic Leukemias
Practice Questions
Lymphomas and Lymphoid Neoplasms
Practice Questions
Plasma Cell Disorders
Practice Questions
Bleeding Disorders
Practice Questions
Thrombotic Disorders
Practice Questions
Want unlimited practice?
Get full access to all questions, explanations, and performance tracking.
Scan to download app
