General Pathology — MCQs

On this page
All of the following are stress sensors/regulators for apoptosis, except?
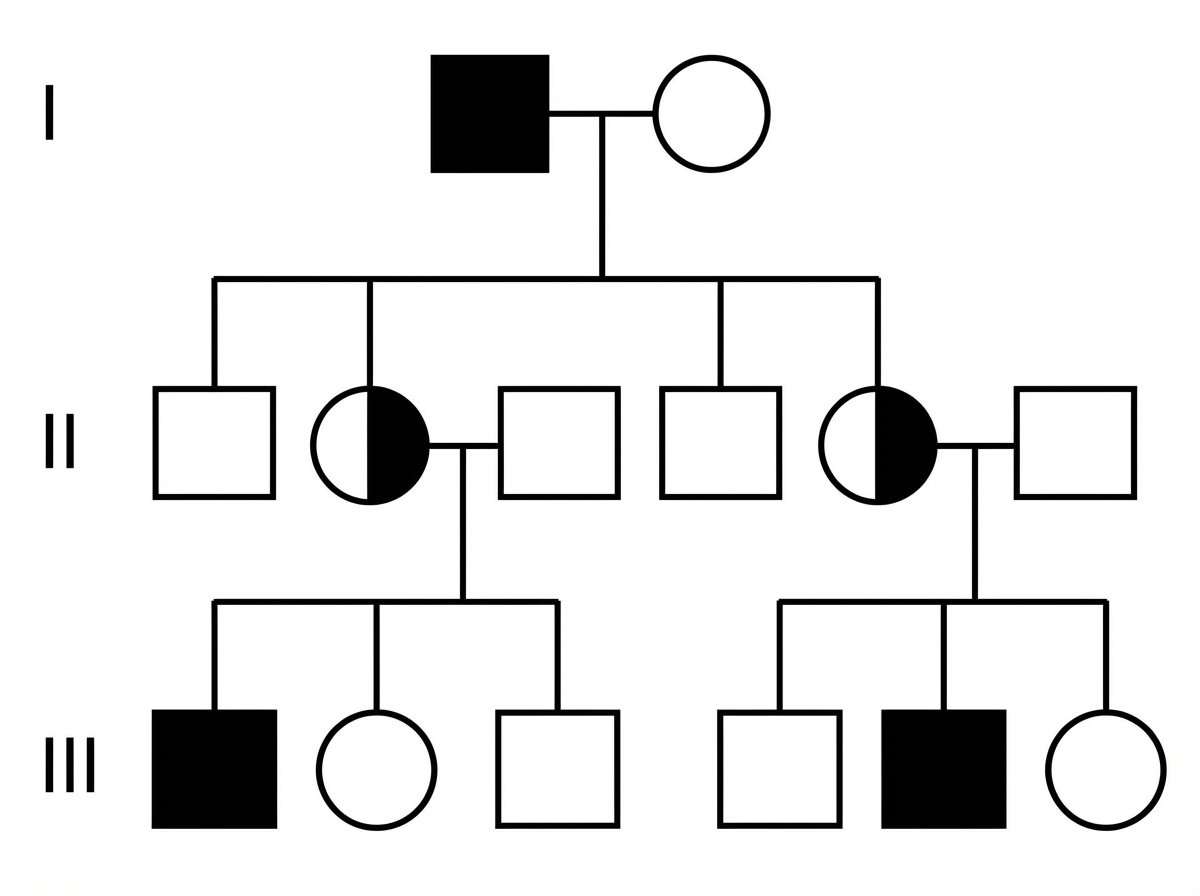
The given pedigree chart most appropriately resembles which of the following diseases?
When should EDTA be added to a blood sample?
Which immunoglobulin is primarily found in mucosal secretions?
Which stain is used for melanin?
Which is the most common complement protein deficiency?
Which of the following statements regarding the Human Leukocyte Antigen (HLA) complex is incorrect?
Which of the following conditions is associated with coagulative necrosis?
Which of the following is characteristic of irreversible cell injury?
Which gene is involved in endometrial carcinoma?
Practice by Chapter
Cell Injury and Cell Death
Practice Questions
Adaptations of Cellular Growth
Practice Questions
Accumulations and Deposits
Practice Questions
Acute and Chronic Inflammation
Practice Questions
Tissue Repair and Wound Healing
Practice Questions
Hemodynamic Disorders
Practice Questions
Genetic Disorders
Practice Questions
Environmental Pathology
Practice Questions
Nutritional Diseases
Practice Questions
Molecular Basis of Disease
Practice Questions
Want unlimited practice?
Get full access to all questions, explanations, and performance tracking.
Scan to download app
