General Pathology — MCQs

On this page
Which of the following statements about Becker muscular dystrophy is FALSE?
All of the following are opsonins, except?
Amyloid material can be best diagnosed by which method?
Yellow or green pigmentation of teeth is seen in which of the following conditions?
Which stain is used for melanin?
Common variable hypogammaglobulinemia shows which of the following?
Secondary amyloidosis is seen most commonly in which of the following conditions?
Which of the following conditions can present with numbness of the lip in the absence of previous dental treatment?
Which proto-oncogene is involved in Gastrointestinal Stromal Tumors (GIST)?
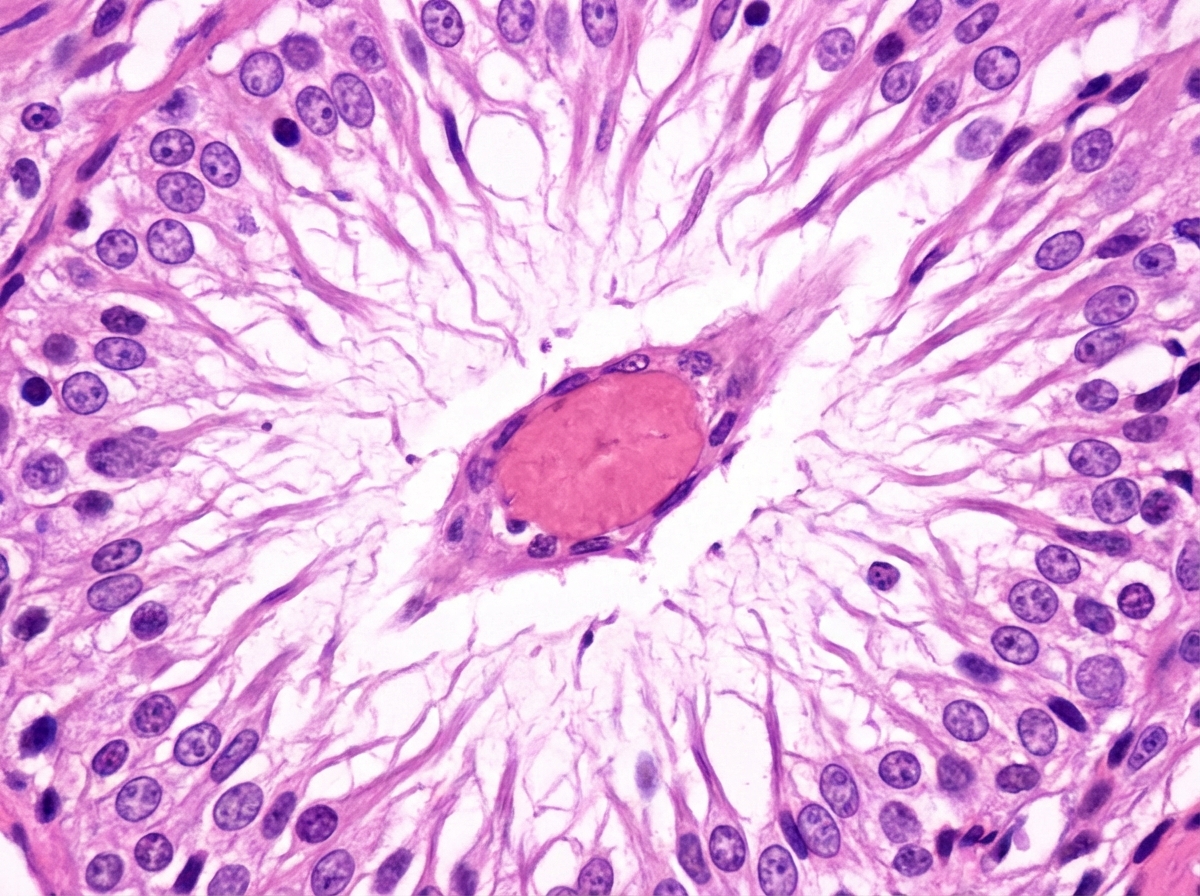
Identify the rosette and its corresponding diagnosis.
Practice by Chapter
Cell Injury and Cell Death
Practice Questions
Adaptations of Cellular Growth
Practice Questions
Accumulations and Deposits
Practice Questions
Acute and Chronic Inflammation
Practice Questions
Tissue Repair and Wound Healing
Practice Questions
Hemodynamic Disorders
Practice Questions
Genetic Disorders
Practice Questions
Environmental Pathology
Practice Questions
Nutritional Diseases
Practice Questions
Molecular Basis of Disease
Practice Questions
Want unlimited practice?
Get full access to all questions, explanations, and performance tracking.
Scan to download app
