General Pathology — MCQs

On this page
Which of the following is FALSE regarding a granuloma?
Fats are stained by which of the following methods?
Which of the following is an example of physiological atrophy?
All of the following can trigger sensors of cell damage in the cytoplasm, except:
Which fixative is commonly used in histopathology?
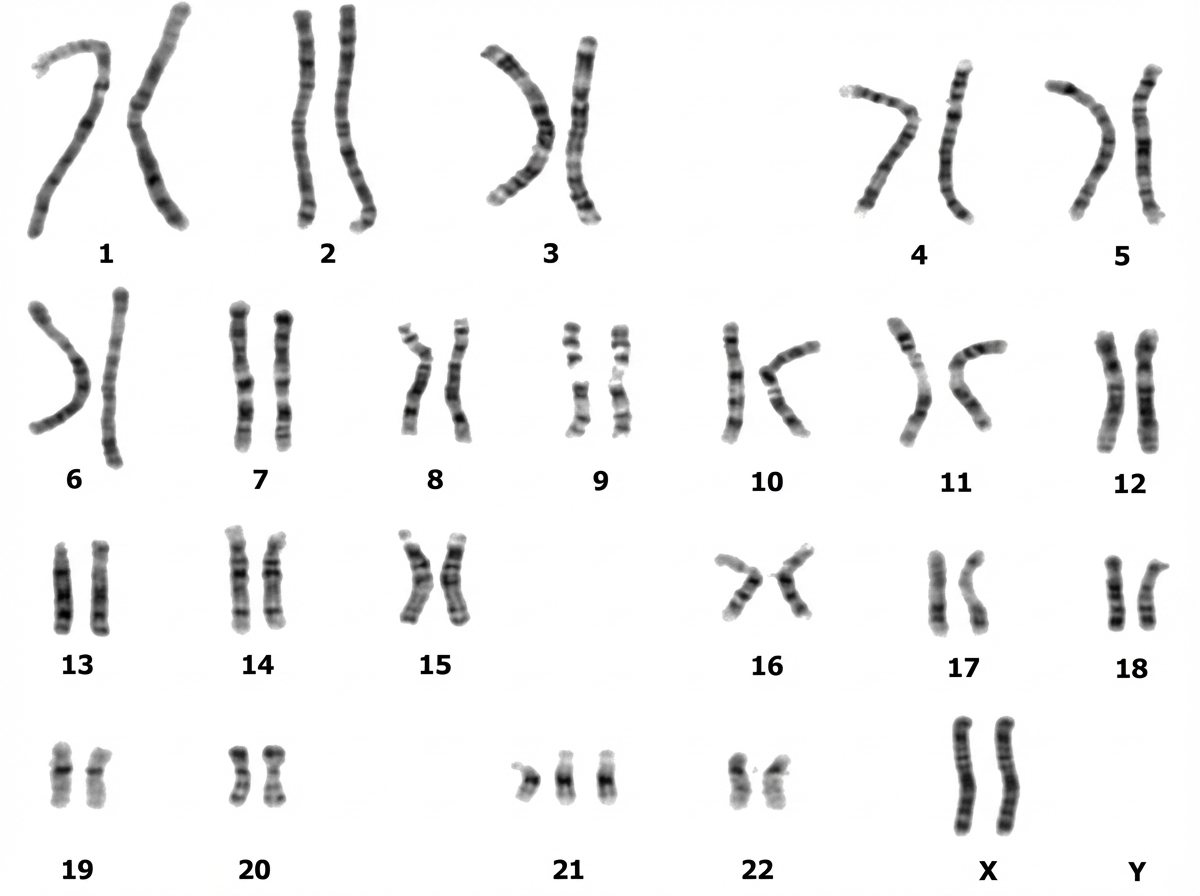
The gene that predisposes to retinoblastoma is located at which chromosomal band?
Irregular opaque areas are found on radiographs of lower anterior teeth. The teeth are vital. What may be the cause?
Green discoloration on the surface of teeth is due to which of the following?
Pyroptosis is associated with which of the following cytokines?
This gene mapping indicates:
Practice by Chapter
Cell Injury and Cell Death
Practice Questions
Adaptations of Cellular Growth
Practice Questions
Accumulations and Deposits
Practice Questions
Acute and Chronic Inflammation
Practice Questions
Tissue Repair and Wound Healing
Practice Questions
Hemodynamic Disorders
Practice Questions
Genetic Disorders
Practice Questions
Environmental Pathology
Practice Questions
Nutritional Diseases
Practice Questions
Molecular Basis of Disease
Practice Questions
Want unlimited practice?
Get full access to all questions, explanations, and performance tracking.
Scan to download app
