General Pathology — MCQs

On this page
Which of the following statements regarding red infarcts is incorrect?
What is the molecular change typically seen in lysosomal storage disorders?
A histopathological image of a cystic lesion is shown. What is the most likely diagnosis?
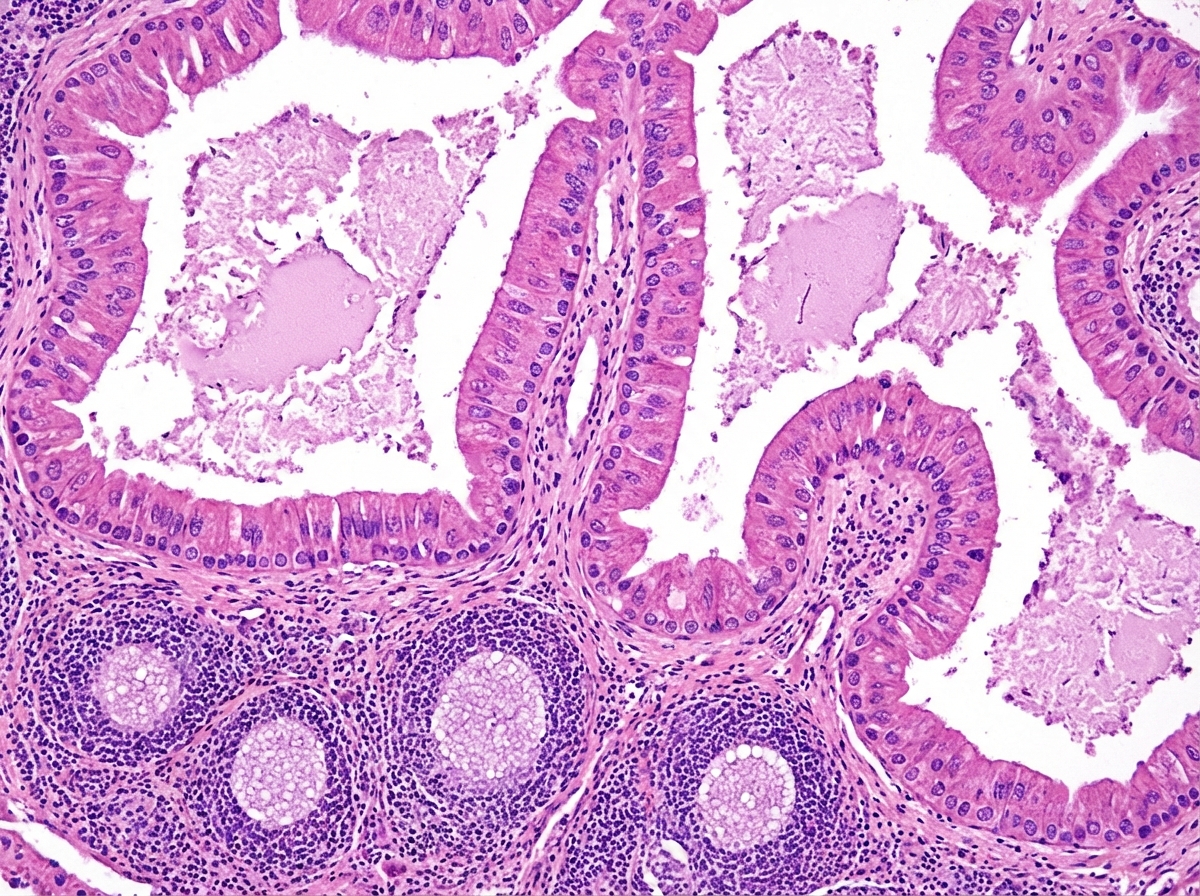
What characterizes a granuloma?
A 24-year-old man is found to have an increased total body iron concentration. Biopsy of his liver shows large amounts of granular golden-brown pigment which stains blue with Prussian blue stain. The presence of which one of the following diseases best explains these findings?
Which of the following is true about stem cells?
Accumulation of which of the following substances causes fatty liver?
What is Turner hypoplasia?
Gene for retinoblastoma is located on which chromosome?
Which of the following is an example of metastatic calcification?
Practice by Chapter
Cell Injury and Cell Death
Practice Questions
Adaptations of Cellular Growth
Practice Questions
Accumulations and Deposits
Practice Questions
Acute and Chronic Inflammation
Practice Questions
Tissue Repair and Wound Healing
Practice Questions
Hemodynamic Disorders
Practice Questions
Genetic Disorders
Practice Questions
Environmental Pathology
Practice Questions
Nutritional Diseases
Practice Questions
Molecular Basis of Disease
Practice Questions
Want unlimited practice?
Get full access to all questions, explanations, and performance tracking.
Scan to download app
