Gastrointestinal Pathology — MCQs

On this page
A small intestinal biopsy is specific for which of the following conditions?
Early gastric carcinoma is indicated by what finding?
The mother of a 4-year-old child notes blood when laundering his underwear. Physical examination reveals a rectal mass. On proctoscopy, there is a smooth-surfaced, pedunculated, 1.5-cm polyp. It is excised and microscopically shows cystically dilated crypts filled with mucin and inflammatory debris, but no dysplasia. What is the most likely diagnosis?
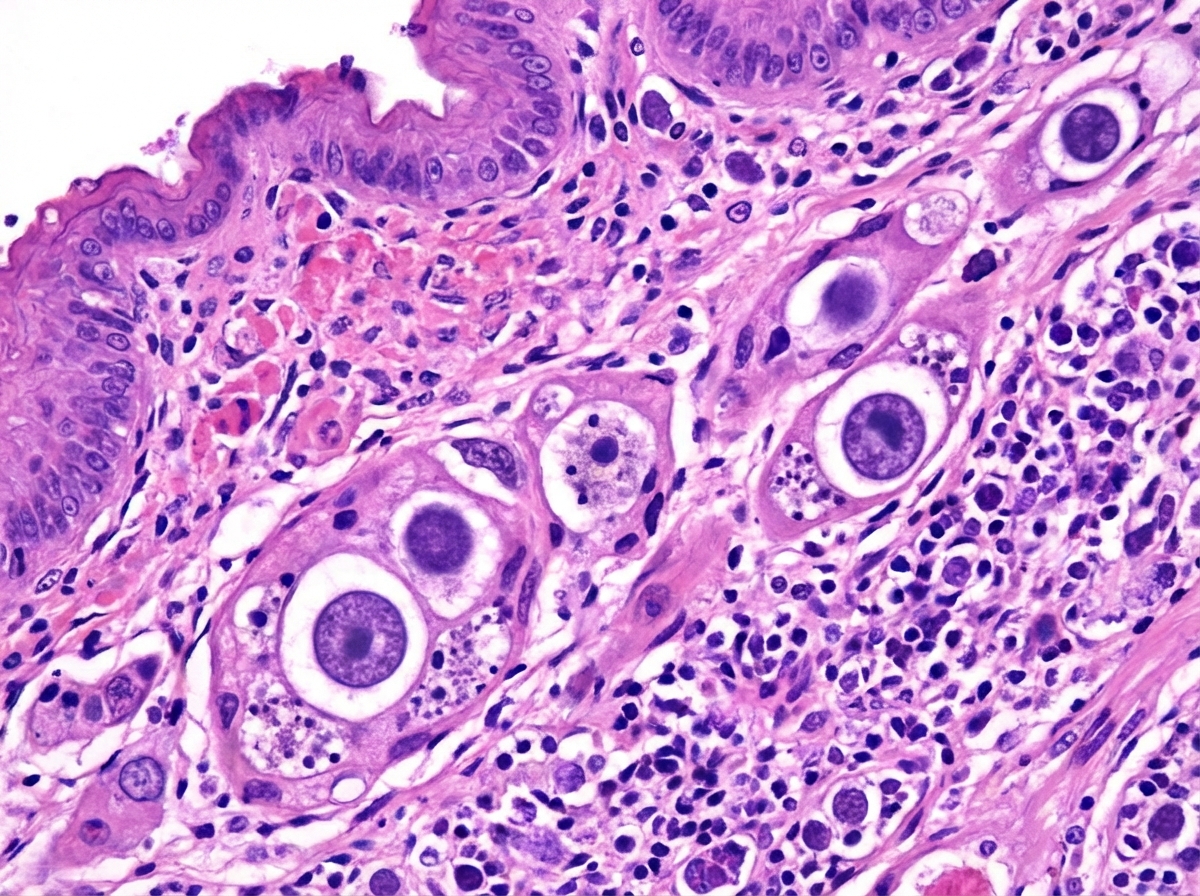
A 40-year-old immunocompromised patient develops multiple ulcers in the esophagus leading to painful swallowing. The biopsy from the esophagus is shown below. What is the diagnosis?
Which of the following statements about ulcerative colitis (UC) and colon carcinoma is FALSE?
Which one of the following conditions commonly predisposes to colonic carcinoma?
Jejunal biopsy is diagnostic in which of the following conditions?
A 70-year-old man presents with fatigue, weight loss, abdominal pain, and blood in the stools. A complete blood count reveals anemia with hemoglobin of 10 g/dL. A colonoscopy and subsequent colonic biopsy reveal adenocarcinoma. Which of the following is the most likely predisposing lesion or disorder that led to this malignancy?
The APC gene is involved in which of the following conditions?
Collar Button Ulcer is a feature of which condition?
Practice by Chapter
Oral Cavity and Esophageal Pathology
Practice Questions
Gastritis and Peptic Ulcer Disease
Practice Questions
Inflammatory Bowel Disease
Practice Questions
Malabsorption Syndromes
Practice Questions
Vascular Disorders of Intestine
Practice Questions
Diverticular Disease
Practice Questions
Intestinal Obstruction
Practice Questions
Gastrointestinal Infections
Practice Questions
Polyps and Neoplasms
Practice Questions
Appendiceal Pathology
Practice Questions
Want unlimited practice?
Get full access to all questions, explanations, and performance tracking.
Scan to download app
