Gastrointestinal Pathology — MCQs

On this page
Which of the following is a true statement about Peutz-Jeghers syndrome?
A 60-year-old male presents with dysphagia. A mucosal biopsy is provided. What does it show?
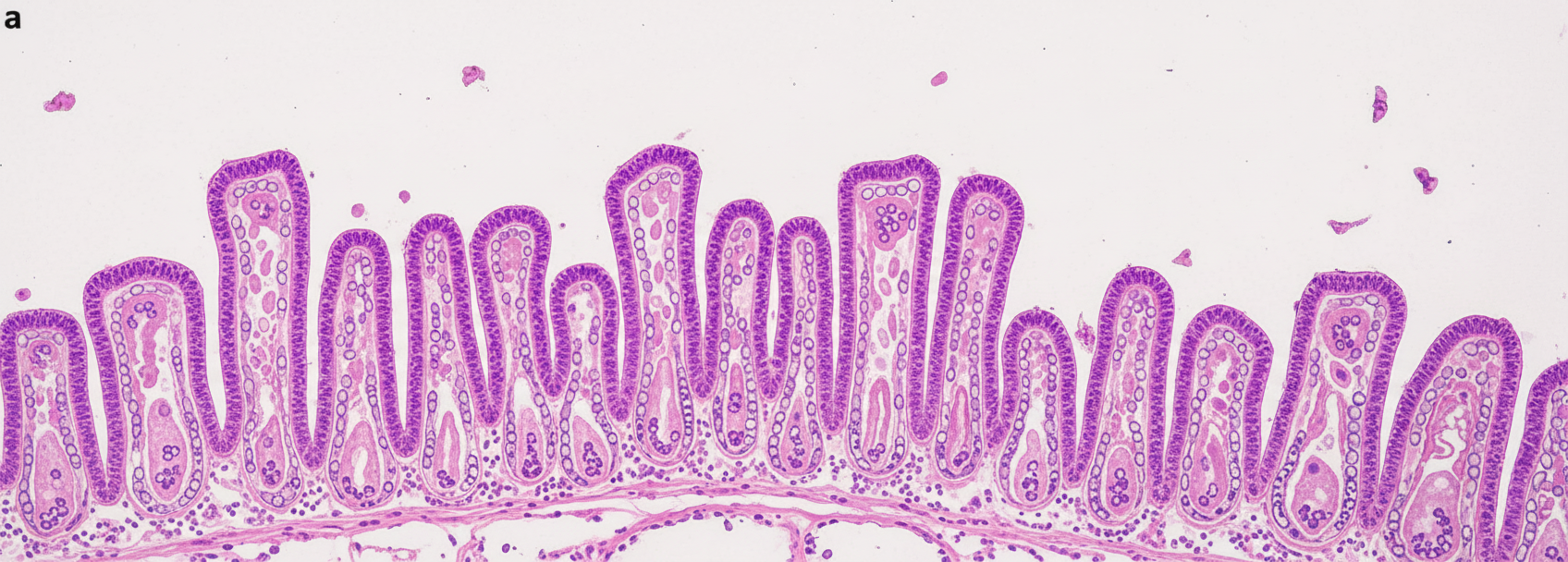
Which of the following is NOT a complication of typhoid ulcers?
What is the etiology of type-A gastritis?
Increased serum amylase may be seen in all of the following conditions except?
What is the most common site of adenocarcinoma in the small intestine?
According to Borrmann's classification, linitis plastica is what type?
What is the most common type of salivary gland neoplasm?
A 14-year-old girl with a history of prolonged fever and abdominal discomfort is observed to have splenomegaly and leucopenia. In the course of the disease, she developed an acute abdominal event and died. Which of the following is the likely finding on autopsy?
Which of the following is NOT a risk factor for gastric cancer?
Practice by Chapter
Oral Cavity and Esophageal Pathology
Practice Questions
Gastritis and Peptic Ulcer Disease
Practice Questions
Inflammatory Bowel Disease
Practice Questions
Malabsorption Syndromes
Practice Questions
Vascular Disorders of Intestine
Practice Questions
Diverticular Disease
Practice Questions
Intestinal Obstruction
Practice Questions
Gastrointestinal Infections
Practice Questions
Polyps and Neoplasms
Practice Questions
Appendiceal Pathology
Practice Questions
Want unlimited practice?
Get full access to all questions, explanations, and performance tracking.
Scan to download app
