Gastrointestinal Pathology — MCQs

On this page
A patient presents with diarrhea, malabsorption, abdominal pain, and arthralgia. Intestinal biopsy reveals PAS-positive organisms within macrophages. What is the most likely diagnosis?
A 50-year-old male presents with a history of Gastroesophageal reflux disease. What is the most common pathological finding associated with this condition?
What is the most common type of gastric polyp?
Which marker is used for the early diagnosis of Hirschsprung disease?
What is the most important prognostic indicator in esophageal carcinoma?
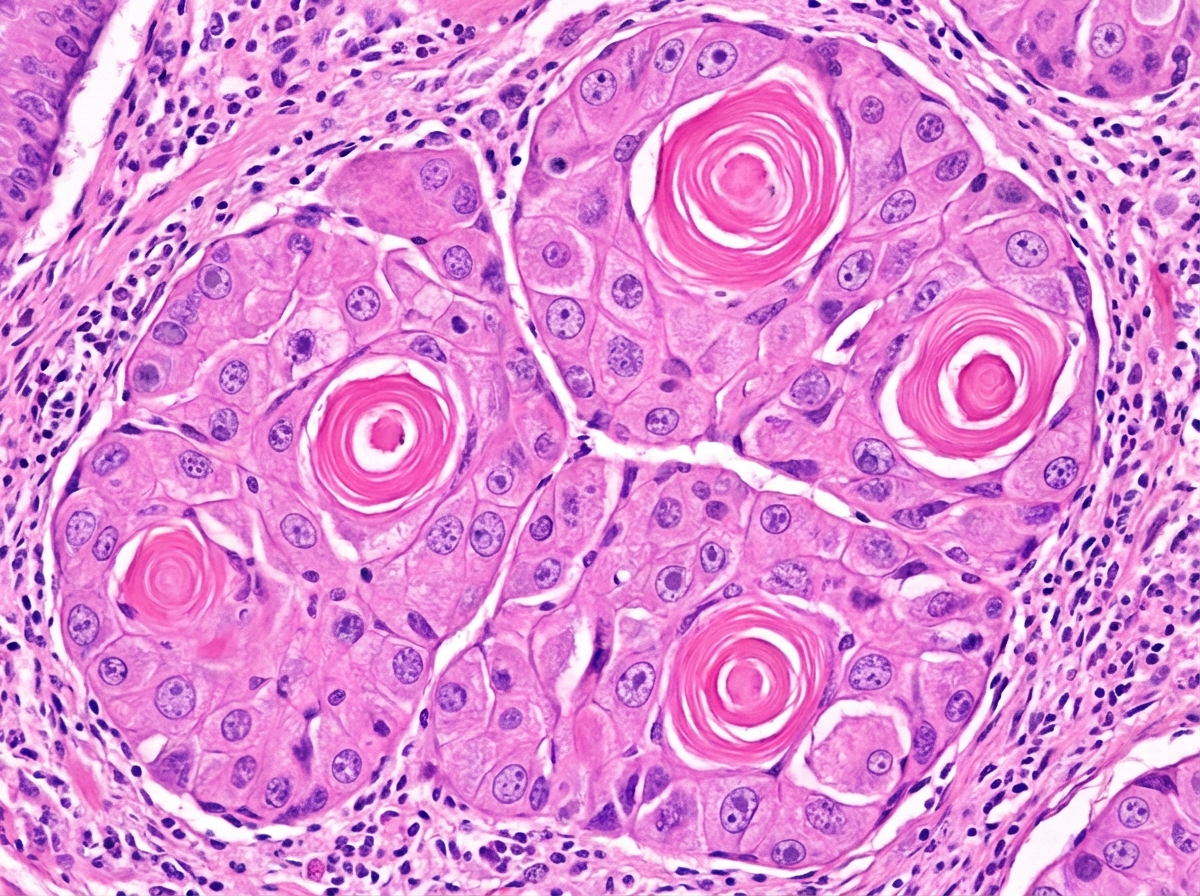
A 60-year-old patient with a prolonged history of tobacco chewing presents with symptoms of weight loss and dysphagia. An esophageal biopsy from a projectile mass is shown below. What is your likely diagnosis?
Meckel Diverticulum is a remnant of which embryonic structure?
What is characteristic of a typhoid ulcer?
Which of the following anaemias is a risk factor for the development of gastric carcinoma?
Type B gastritis is characterized by predominance in which region of the stomach?
Practice by Chapter
Oral Cavity and Esophageal Pathology
Practice Questions
Gastritis and Peptic Ulcer Disease
Practice Questions
Inflammatory Bowel Disease
Practice Questions
Malabsorption Syndromes
Practice Questions
Vascular Disorders of Intestine
Practice Questions
Diverticular Disease
Practice Questions
Intestinal Obstruction
Practice Questions
Gastrointestinal Infections
Practice Questions
Polyps and Neoplasms
Practice Questions
Appendiceal Pathology
Practice Questions
Want unlimited practice?
Get full access to all questions, explanations, and performance tracking.
Scan to download app
