Endocrine Pathology — MCQs

On this page
What is the most common cause of primary hyperparathyroidism?
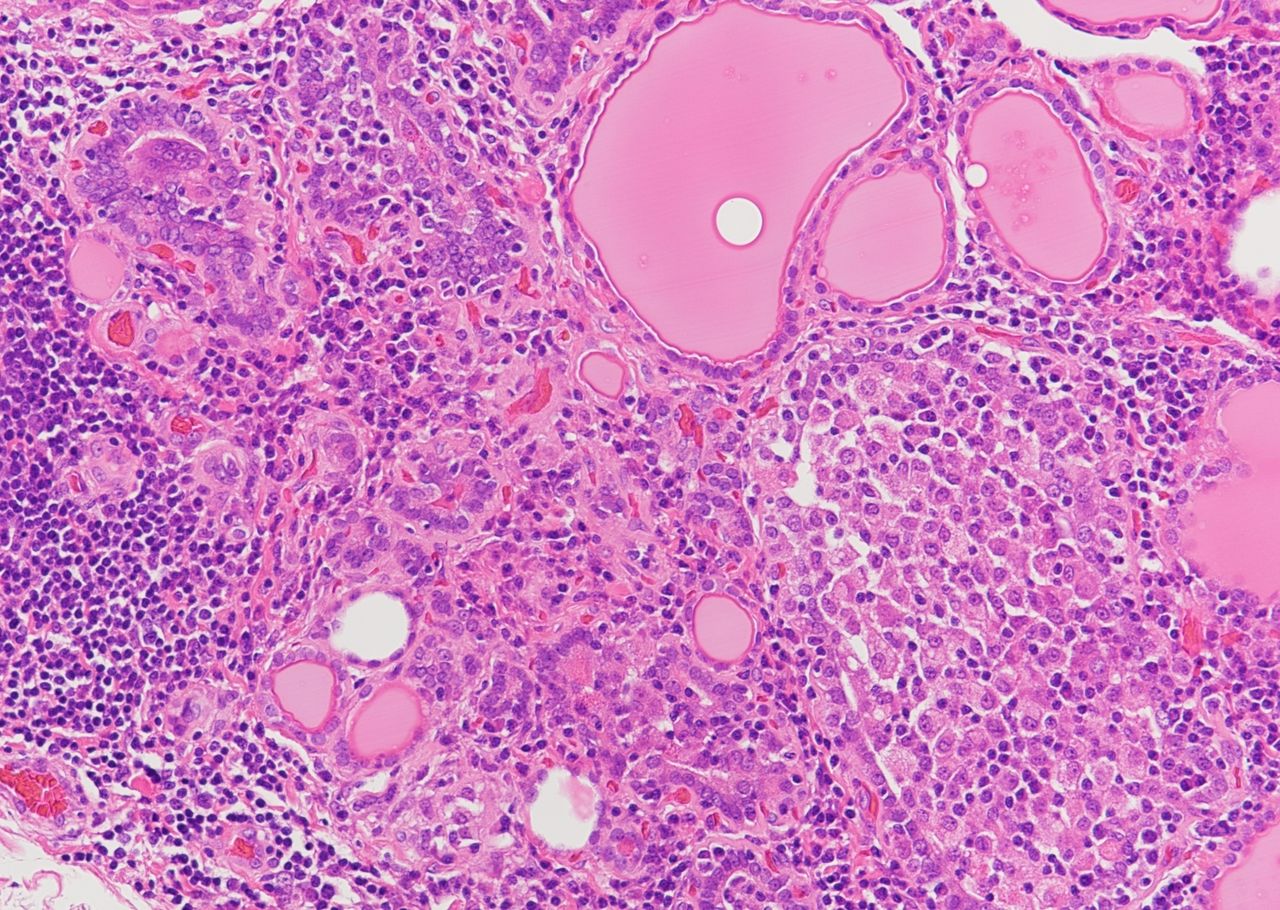
A 45-year-old female patient presented with features of hypothyroidism. Based on the provided histological features, what is your diagnosis?
All are seen in MEN IIA syndrome except?
Pancreatitis, pituitary tumor, and pheochromocytoma may be associated with which of the following types of thyroid cancer?
Carcinoid tumours commonly arise from which cell type and location?
Pheochromocytomas are known to arise from all of the following locations, except:
All of the following are seen in follicular carcinoma of the thyroid, except?
Crook's hyaline change occurs in which of the following?
A female neonate with DiGeorge syndrome develops severe muscle cramps and convulsions soon after birth. Which of the following is the cause of convulsions in this neonate?
Hürthle cell carcinoma is a variant of which thyroid carcinoma?
Practice by Chapter
Pituitary Gland Disorders
Practice Questions
Thyroid Gland Diseases
Practice Questions
Parathyroid Gland Pathology
Practice Questions
Adrenal Cortical Disorders
Practice Questions
Adrenal Medullary Disorders
Practice Questions
Pancreatic Endocrine Disorders
Practice Questions
Multiple Endocrine Neoplasia Syndromes
Practice Questions
Diffuse Neuroendocrine System
Practice Questions
Pineal Gland Pathology
Practice Questions
Laboratory Diagnosis of Endocrine Diseases
Practice Questions
Want unlimited practice?
Get full access to all questions, explanations, and performance tracking.
Scan to download app
