Endocrine Pathology — MCQs

On this page
Medullary carcinoma of the thyroid arises from which of the following cells?
What is true about papillary carcinoma?
Which type of adenoma has the highest propensity to undergo dystrophic calcification?
A 29-year-old woman presents with a 6-month history of nervousness, muscle weakness, heat intolerance, excessive sweating, and a 9 kg weight loss despite increased caloric intake. She also reports palpitations and amenorrhea. Physical examination reveals warm, moist skin and exophthalmos. A thyroid biopsy is performed. Which of the following best describes the expected pathologic findings?
Pheochromocytoma with malignant potential exclusively secretes which of the following?
In malignant hyperthermia, what causes the increased heat production?
Familial hypocalciuric hypercalcemia is characterized by mild elevation of calcium and parathyroid hormone (PTH) levels. It is primarily caused by a mutation in which of the following?
A 58-year-old woman presents with difficulty in swallowing. After a comprehensive workup by a gastroenterologist rules out primary esophageal disease, she is referred to an endocrinologist. The endocrinologist suspects Riedel thyroiditis. Which of the following physical examination findings would best help confirm this diagnosis?
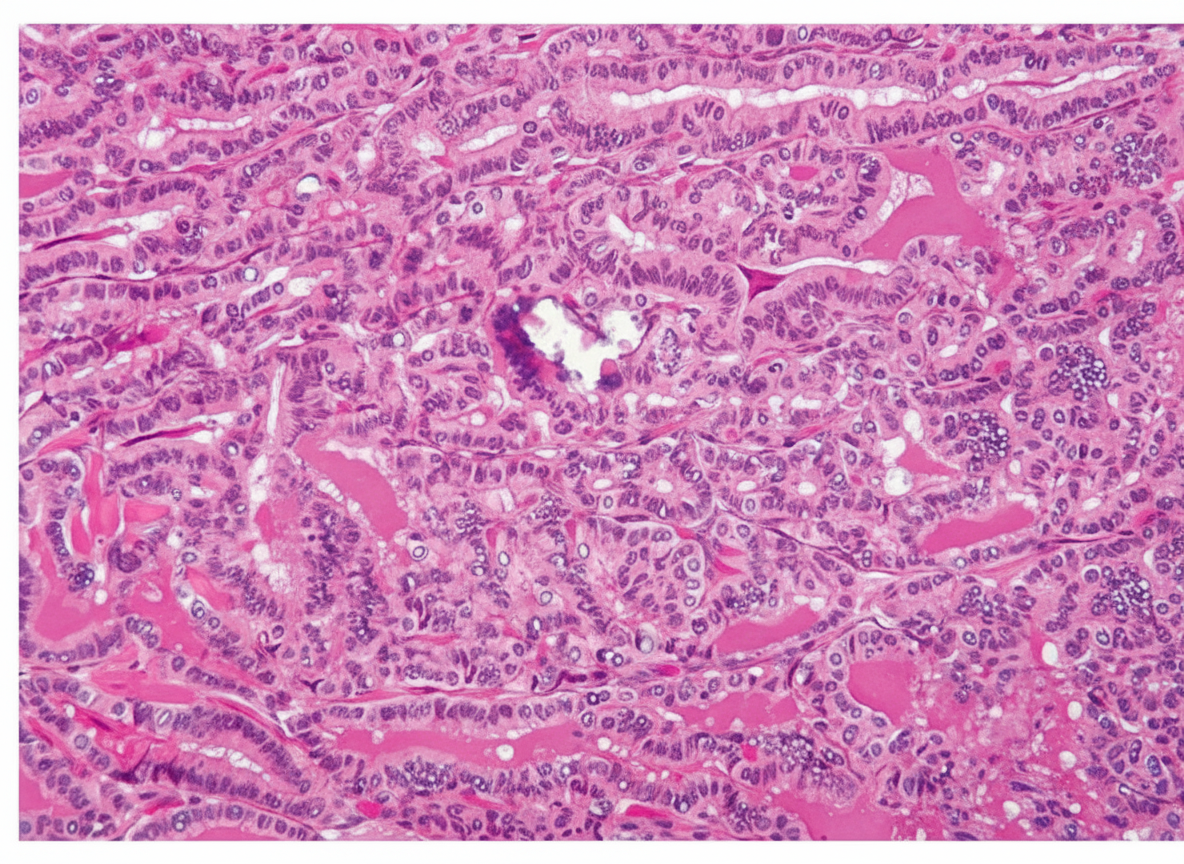
A tumor similar to that shown in the illustration is observed in a biopsy specimen from the thyroid of a 50-year-old woman. An adjacent lymph node is also involved. Which of the following descriptions of this tumor is most appropriate?
Thymic hyperplasia is seen in which condition?
Practice by Chapter
Pituitary Gland Disorders
Practice Questions
Thyroid Gland Diseases
Practice Questions
Parathyroid Gland Pathology
Practice Questions
Adrenal Cortical Disorders
Practice Questions
Adrenal Medullary Disorders
Practice Questions
Pancreatic Endocrine Disorders
Practice Questions
Multiple Endocrine Neoplasia Syndromes
Practice Questions
Diffuse Neuroendocrine System
Practice Questions
Pineal Gland Pathology
Practice Questions
Laboratory Diagnosis of Endocrine Diseases
Practice Questions
Want unlimited practice?
Get full access to all questions, explanations, and performance tracking.
Scan to download app
