Endocrine Pathology — MCQs

On this page
Medullary thyroid carcinoma occurs due to the mutation of which oncogene or gene?
All of the following are features of De Quervain thyroiditis except?
Multiple Endocrine Neoplasia (MEN) types II and III are associated with which gene mutation?
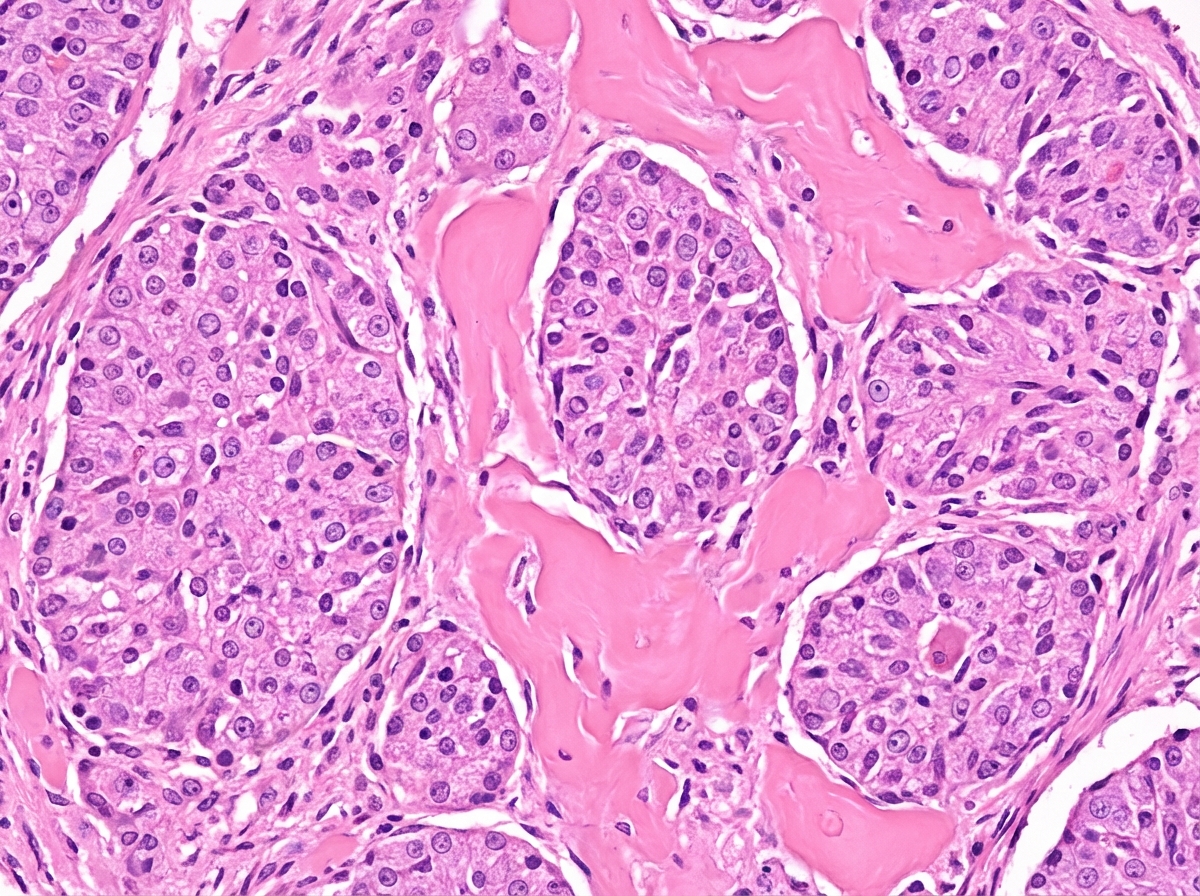
A 40-year-old male presented with a thyroid swelling and dysphagia. He gave history of on and off watery diarrhea. Biopsy of the lesion is shown. What is your diagnosis?
Which of the following is the most reliable feature of malignant transformation of pheochromocytoma?
Which of the following is seen on electron microscopy of medullary thyroid carcinoma specimens?
What is the ultrastructural finding in a case of paraganglioma?
A 21-year-old woman experiences abruptio placentae with severe bleeding during the delivery of a term fetus. Five months later, she presents with profound lethargy, pallor, muscle weakness, failure of lactation, and amenorrhea. Which of the following pathologic findings is expected in this patient?
All of the following are TRUE about Hashimoto's Thyroiditis, EXCEPT?
Spironolactone bodies are seen in which cellular structure?
Practice by Chapter
Pituitary Gland Disorders
Practice Questions
Thyroid Gland Diseases
Practice Questions
Parathyroid Gland Pathology
Practice Questions
Adrenal Cortical Disorders
Practice Questions
Adrenal Medullary Disorders
Practice Questions
Pancreatic Endocrine Disorders
Practice Questions
Multiple Endocrine Neoplasia Syndromes
Practice Questions
Diffuse Neuroendocrine System
Practice Questions
Pineal Gland Pathology
Practice Questions
Laboratory Diagnosis of Endocrine Diseases
Practice Questions
Want unlimited practice?
Get full access to all questions, explanations, and performance tracking.
Scan to download app
