Endocrine Pathology — MCQs

On this page
Which of the following mechanisms is NOT responsible for complications in Diabetes Mellitus?
Pheochromocytoma are tumors of which structure?
Expression of which of the following oncogenes is associated with a high incidence of medullary carcinoma of the thyroid?
A biopsy from the parathyroid gland of a 55-year-old male, a known case of chronic kidney disease with hypertension and type II diabetes who recently developed bone pain, skin lesions, and recurrent kidney stones, is shown below. What is the most likely histopathological finding?
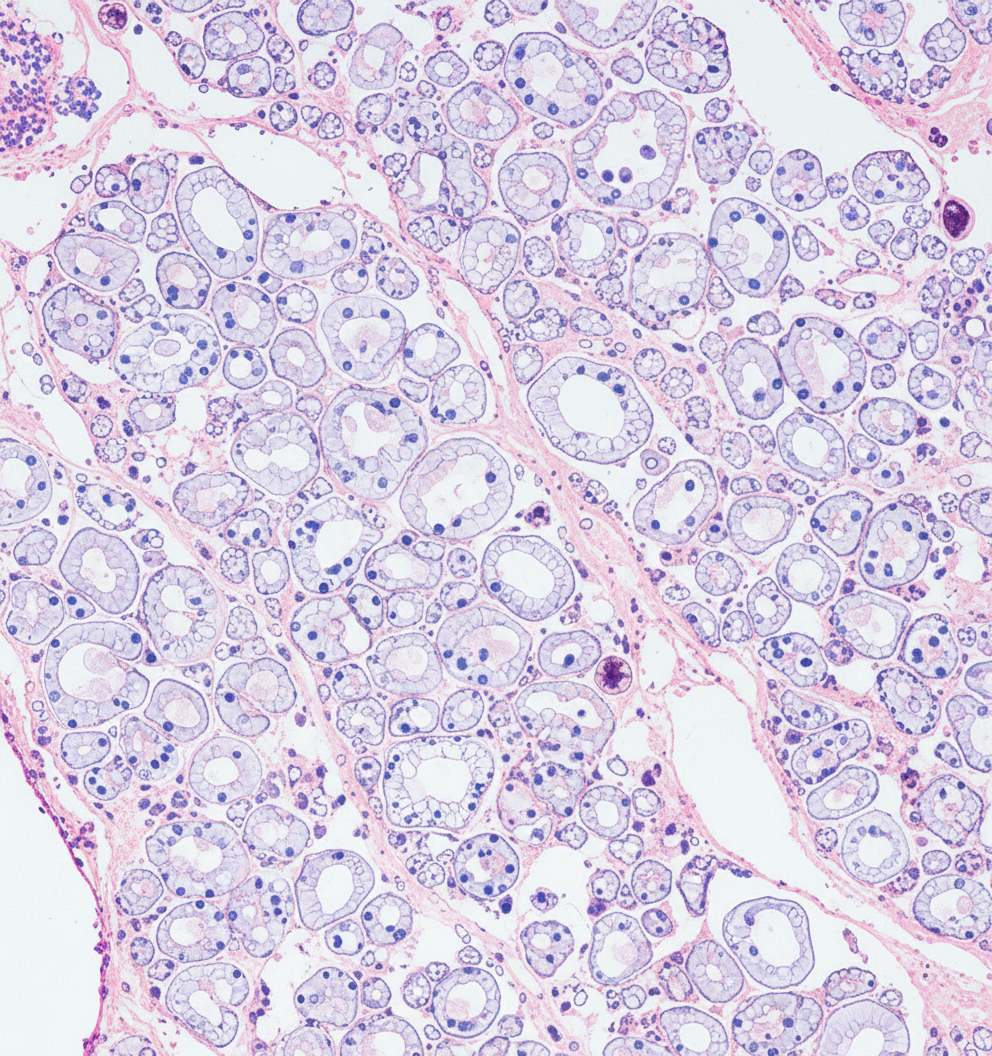
The neuroendocrine carcinoma arising from parafollicular 'C' cells of thyroid is:
A 30-year-old woman presents with headache, visual disturbances, deepening of the voice, generalized weakness, amenorrhea for one year, and a recent requirement for a larger shoe size. Laboratory studies show impaired glucose tolerance. What additional diagnostic procedure would be most useful?
A 50-year-old man with fasting blood glucose >140 mg/dL on two occasions is put on a restricted caloric diet and started on a glucagon-like peptide-1 (GLP-1) receptor agonist. Which of the following laboratory studies is most likely to afford the best method of monitoring disease control in this man?
Psammoma bodies are typically found in which of the following conditions?
Hyaline in islets of Langerhans resembles which substance?
Which of the following gene defects is associated with the development of medullary carcinoma of the thyroid?
Practice by Chapter
Pituitary Gland Disorders
Practice Questions
Thyroid Gland Diseases
Practice Questions
Parathyroid Gland Pathology
Practice Questions
Adrenal Cortical Disorders
Practice Questions
Adrenal Medullary Disorders
Practice Questions
Pancreatic Endocrine Disorders
Practice Questions
Multiple Endocrine Neoplasia Syndromes
Practice Questions
Diffuse Neuroendocrine System
Practice Questions
Pineal Gland Pathology
Practice Questions
Laboratory Diagnosis of Endocrine Diseases
Practice Questions
Want unlimited practice?
Get full access to all questions, explanations, and performance tracking.
Scan to download app
