Cardiac Pathology — MCQs

On this page
A patient with long-standing, moderately severe anemia dies in an automobile accident. An autopsy is performed. Which of the following cardiac changes will MOST likely be seen when the heart is examined?
What is the most common benign heart tumor?
Which type of endocarditis typically presents with vegetations on both sides of the valves?
What is the pathological finding that results in a tigereye appearance of the heart?
Ascoff's bodies are seen in which condition?
Rupture of the myocardium typically occurs within what time frame after a myocardial infarction?
In a patient with myocardial infarction, which valvular lesion is commonly seen?
Rheumatic activity involves which valves most commonly?
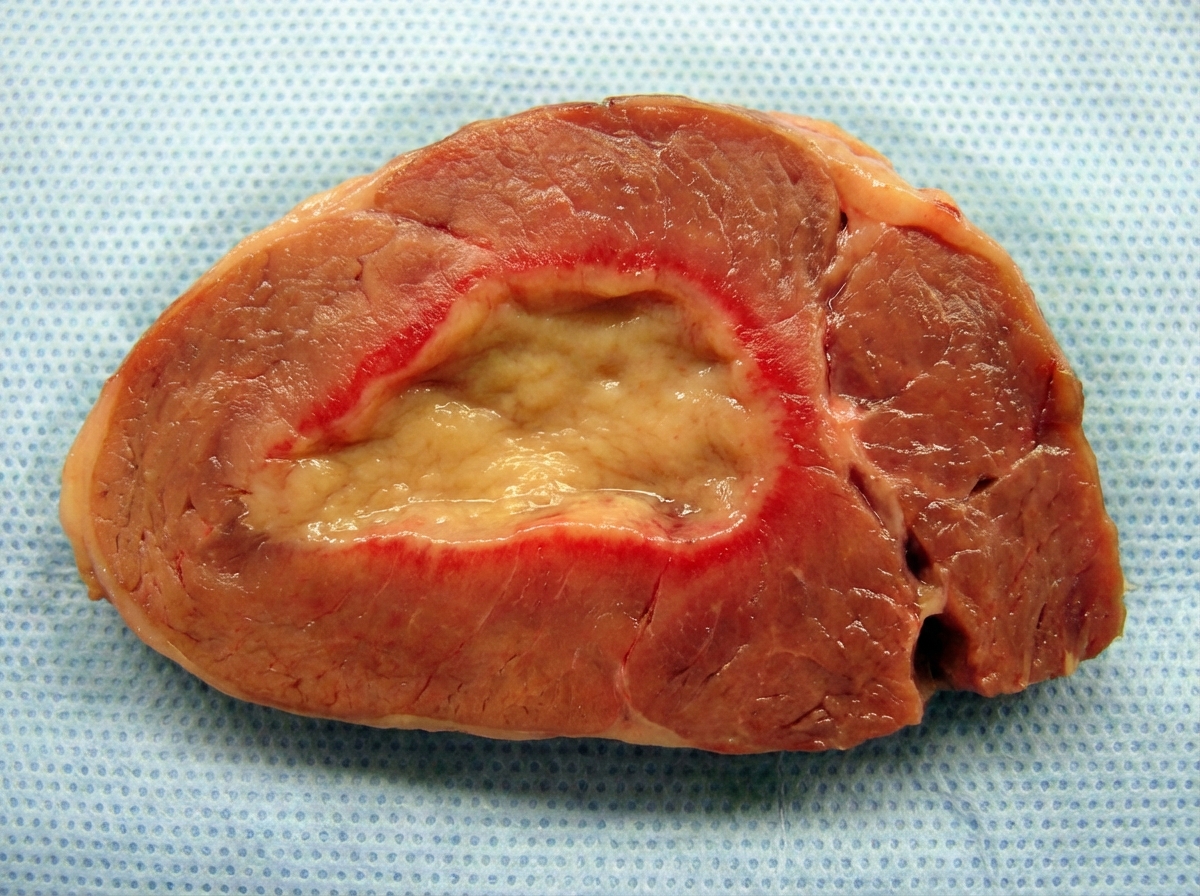
A gross section of the myocardium following myocardial infarction is shown. What is the likely duration following MI?
All are true about myxomas in the heart, EXCEPT:
Practice by Chapter
Congenital Heart Disease
Practice Questions
Ischemic Heart Disease
Practice Questions
Hypertensive Heart Disease
Practice Questions
Valvular Heart Disease
Practice Questions
Myocarditis and Cardiomyopathies
Practice Questions
Pericardial Disease
Practice Questions
Cardiac Tumors
Practice Questions
Heart Failure Pathophysiology
Practice Questions
Cardiac Transplantation Pathology
Practice Questions
Endocarditis
Practice Questions
Want unlimited practice?
Get full access to all questions, explanations, and performance tracking.
Scan to download app
