Cardiac Pathology — MCQs

On this page
Calcification of the aortic valve is seen in which of the following conditions?
A 66-year-old woman collapses and expires suddenly of cardiac arrest. Her past medical history is significant for long-standing type 2 diabetes mellitus. Her relatives note that she had complained of chest heaviness and shortness of breath for the past 2 weeks. Sterile fibrinous pericarditis and pericardial effusion are observed at autopsy. What additional finding would be expected during autopsy of this patient?
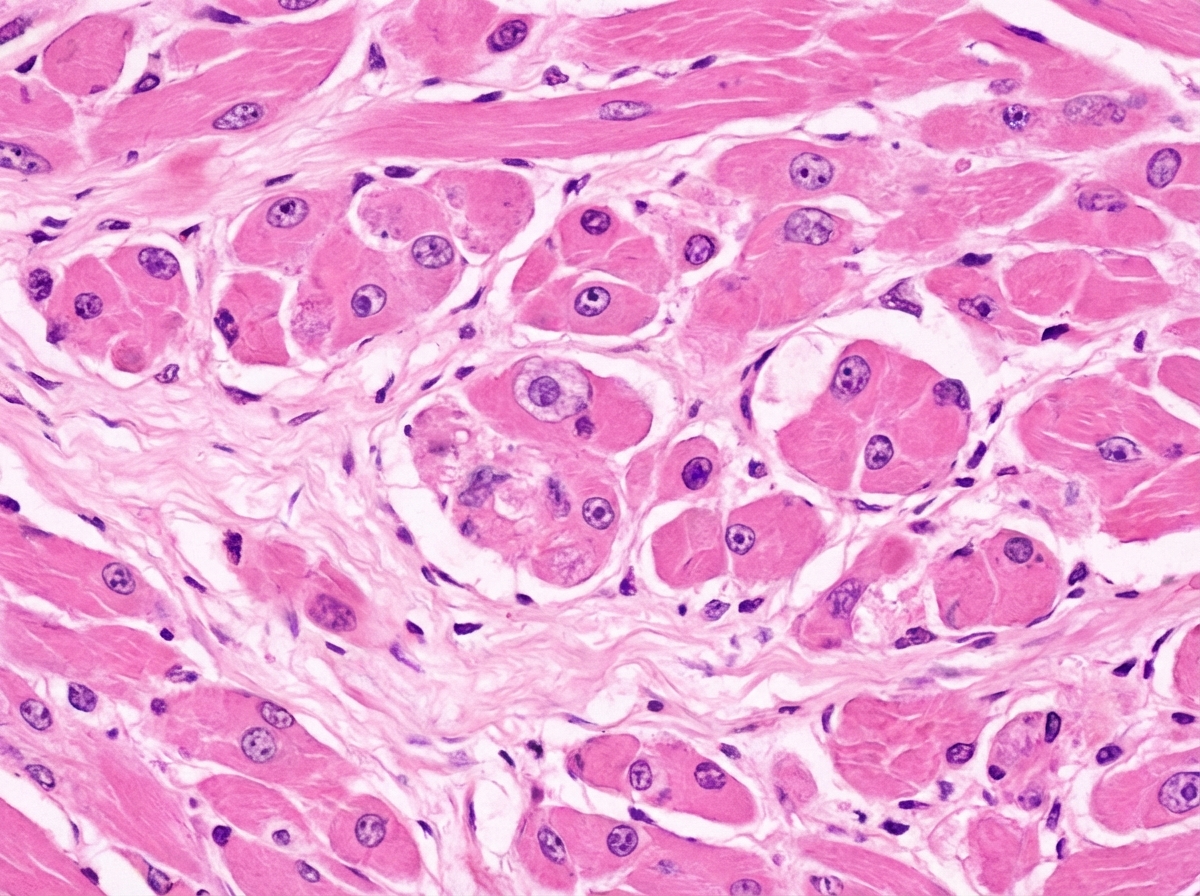
A 25-year-old bodybuilder was using anabolic steroids and started having puffy feet for the last 2 months. He died suddenly during a workout. A post-mortem heart biopsy specimen is shown below. What is the diagnosis?
What is the most common tumor of the heart?
A 59-year-old man experiences acute chest pain and is rushed to the emergency room. Laboratory studies and ECG demonstrate an acute myocardial infarction; however, coronary angiography performed 2 hours later does not show evidence of thrombosis. Which of the following mediators of inflammation causes relaxation of vascular smooth muscle cells and vasodilation of arterioles at the site of myocardial infarction in this patient?
Carcinoid of the heart typically involves which structure?
A 29-year-old male athlete suddenly collapsed and died during a football game. Autopsy revealed a specific gross examination finding. He had a history of two similar deaths in the family. What is the most likely cause of death?
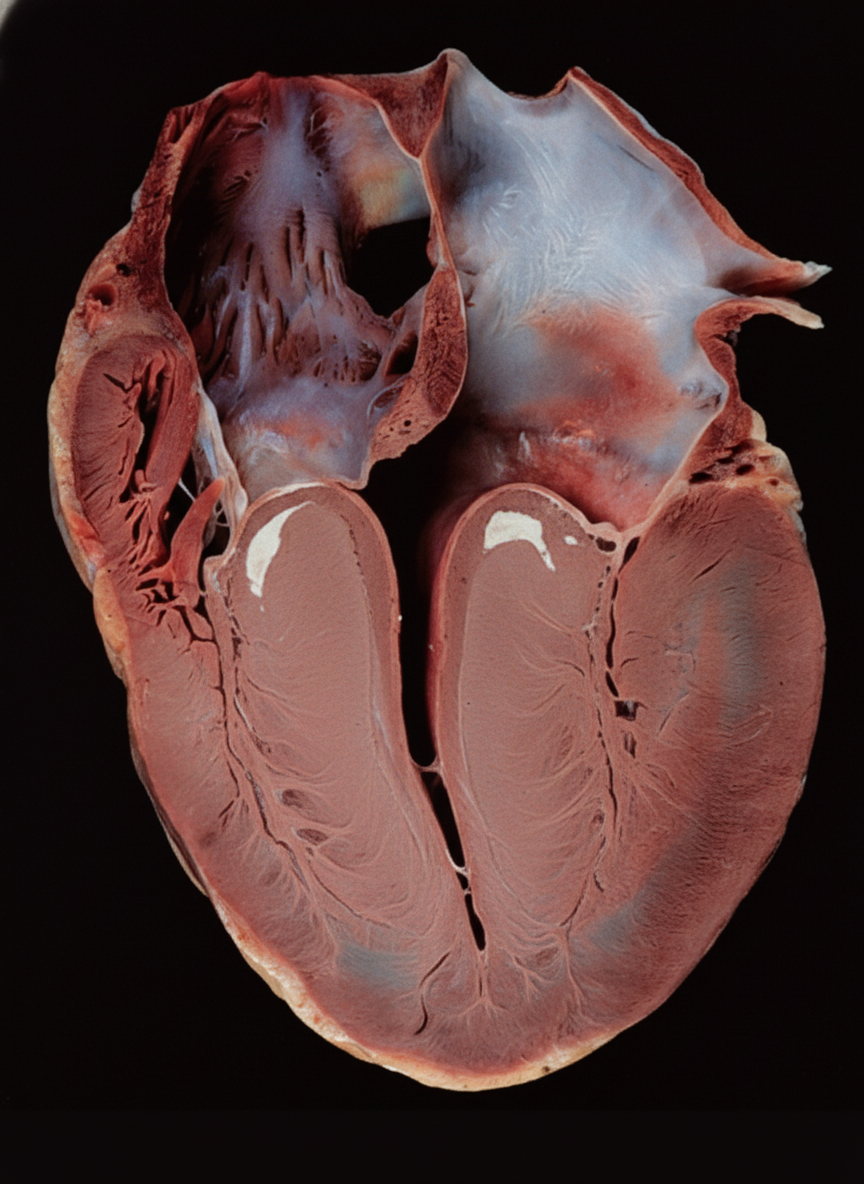
Dilated cardiomyopathy is due to which of the following?
Which of the following dissection methods of the heart is most useful for demonstrating the distribution of infarction?
A 50-year-old woman presents with fatigue and shortness of breath. Physical examination shows evidence of pulmonary edema, enlargement of the left atrium, and calcification of the mitral valve. A CT scan demonstrates a large obstructing mass in the left atrium. Before open heart surgery can be performed to remove the tumor, the patient suffers a stroke and expires. Which of the following hemodynamic disorders best explains the pathogenesis of stroke in this patient?
Practice by Chapter
Congenital Heart Disease
Practice Questions
Ischemic Heart Disease
Practice Questions
Hypertensive Heart Disease
Practice Questions
Valvular Heart Disease
Practice Questions
Myocarditis and Cardiomyopathies
Practice Questions
Pericardial Disease
Practice Questions
Cardiac Tumors
Practice Questions
Heart Failure Pathophysiology
Practice Questions
Cardiac Transplantation Pathology
Practice Questions
Endocarditis
Practice Questions
Want unlimited practice?
Get full access to all questions, explanations, and performance tracking.
Scan to download app
