Cardiac Pathology — MCQs

On this page
What is the most common cause of death in primary amyloidosis?
Which of the following can cause myocarditis?
Mac Callum plaques in rheumatic heart disease are associated with which chamber?
Which is the first organ to be affected in left ventricular failure?
What are the causes of restrictive cardiomyopathy?
What is the earliest time after myocardial infarction when triphenyl tetrazolium chloride (TTC) staining can detect infarcted tissue?
A 65-year-old man dies due to myocardial infarction. Which stains can be used to see the infarct in the heart while conducting an autopsy?
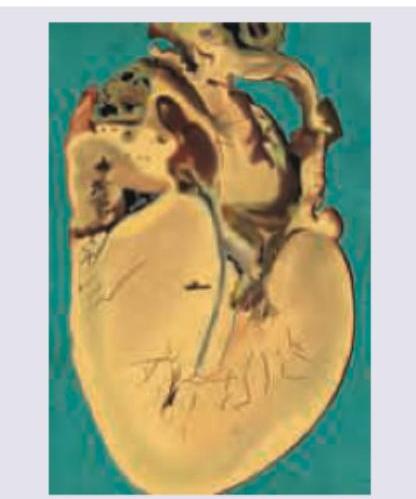
An athlete collapsed suddenly during exercise and died on the field. Postmortem heart is shown in the figure. There is family history of heart disease. What is the diagnosis?
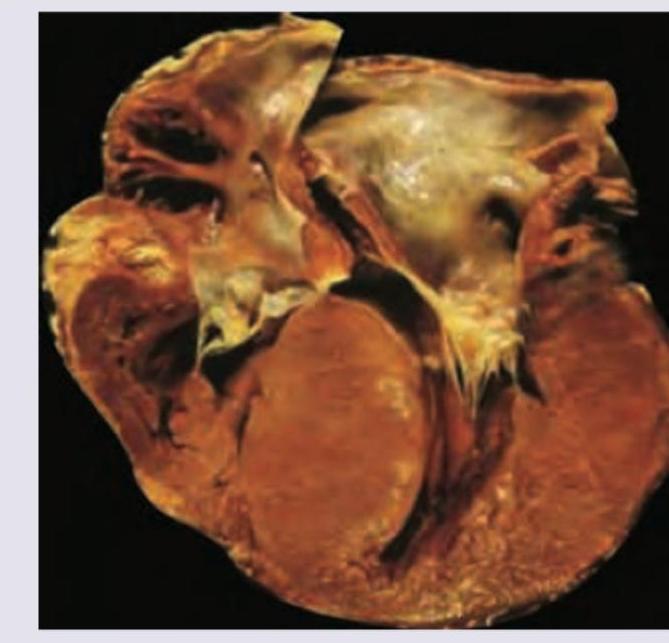
All are true regarding the heart specimen shown except:
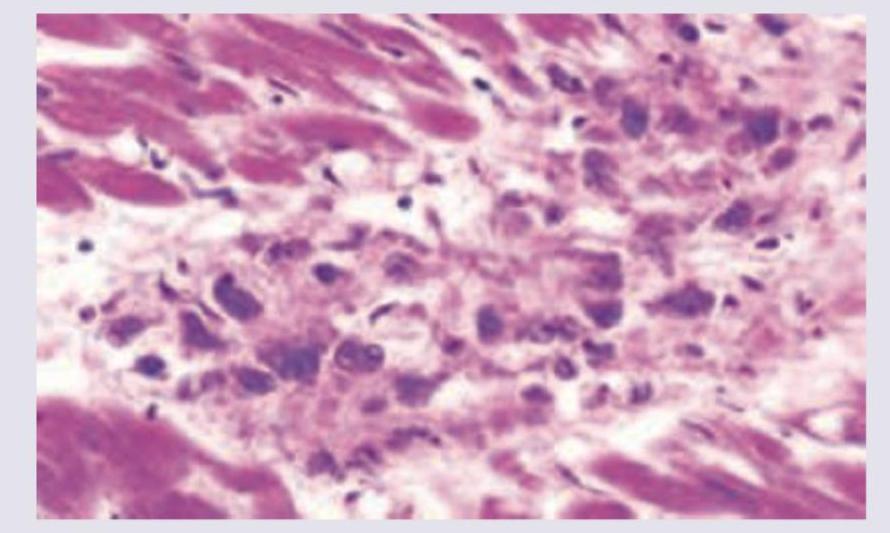
A patient presented with mitral valve stenosis, identify the diagnosis from the mitral valve histopathology depicted below. (AIIMS Nov 2017)
Practice by Chapter
Congenital Heart Disease
Practice Questions
Ischemic Heart Disease
Practice Questions
Hypertensive Heart Disease
Practice Questions
Valvular Heart Disease
Practice Questions
Myocarditis and Cardiomyopathies
Practice Questions
Pericardial Disease
Practice Questions
Cardiac Tumors
Practice Questions
Heart Failure Pathophysiology
Practice Questions
Cardiac Transplantation Pathology
Practice Questions
Endocarditis
Practice Questions
Want unlimited practice?
Get full access to all questions, explanations, and performance tracking.
Scan to download app
