Cardiac Pathology — MCQs

On this page
Which of the following causes the 'no-reflow' phenomenon in acute myocardial infarction, contributing to reperfusion injury?
Which of the following is not commonly associated with non-bacterial thrombotic endocarditis (NBTE)?
A 17-year-old high school student dies suddenly while playing basketball. Autopsy and histologic sections are provided. What is the most likely diagnosis?
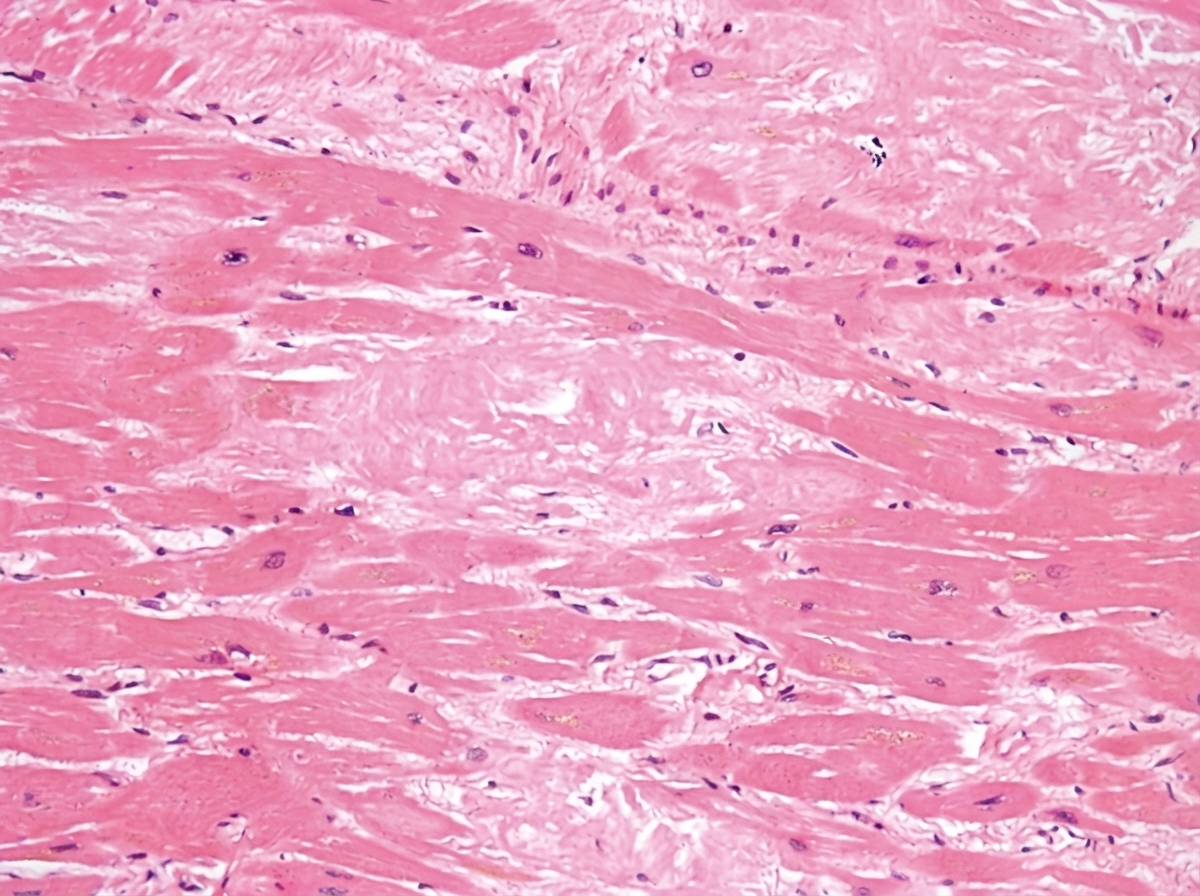
In essential hypertension, what cardiac changes are typically observed?
Heart failure cells are present in which of the following locations?
All of the following statements regarding subendocardial infarction are true, except?
Metastasis to the heart is most commonly from which primary site?
A 25-year-old male presented with a growth in the left atrium. What is the most likely diagnosis?
Calcification of the aortic valve is seen in which of the following conditions?
Hemorrhagic pericarditis occurs in all of the following conditions except?
Practice by Chapter
Congenital Heart Disease
Practice Questions
Ischemic Heart Disease
Practice Questions
Hypertensive Heart Disease
Practice Questions
Valvular Heart Disease
Practice Questions
Myocarditis and Cardiomyopathies
Practice Questions
Pericardial Disease
Practice Questions
Cardiac Tumors
Practice Questions
Heart Failure Pathophysiology
Practice Questions
Cardiac Transplantation Pathology
Practice Questions
Endocarditis
Practice Questions
Want unlimited practice?
Get full access to all questions, explanations, and performance tracking.
Scan to download app
