Cardiac Pathology — MCQs

On this page
Infarcts involving which part of the myocardium cause aneurysm as a post-myocardial infarction complication?
What is the characteristic cardiac lesion in SLE?
Aschoff bodies in Rheumatic heart disease show all of the following features, except?
Troponin T is a marker of which of the following conditions?
What is the most common type of pericarditis seen in tuberculosis?
Concentric hypertrophy of the heart is caused by:
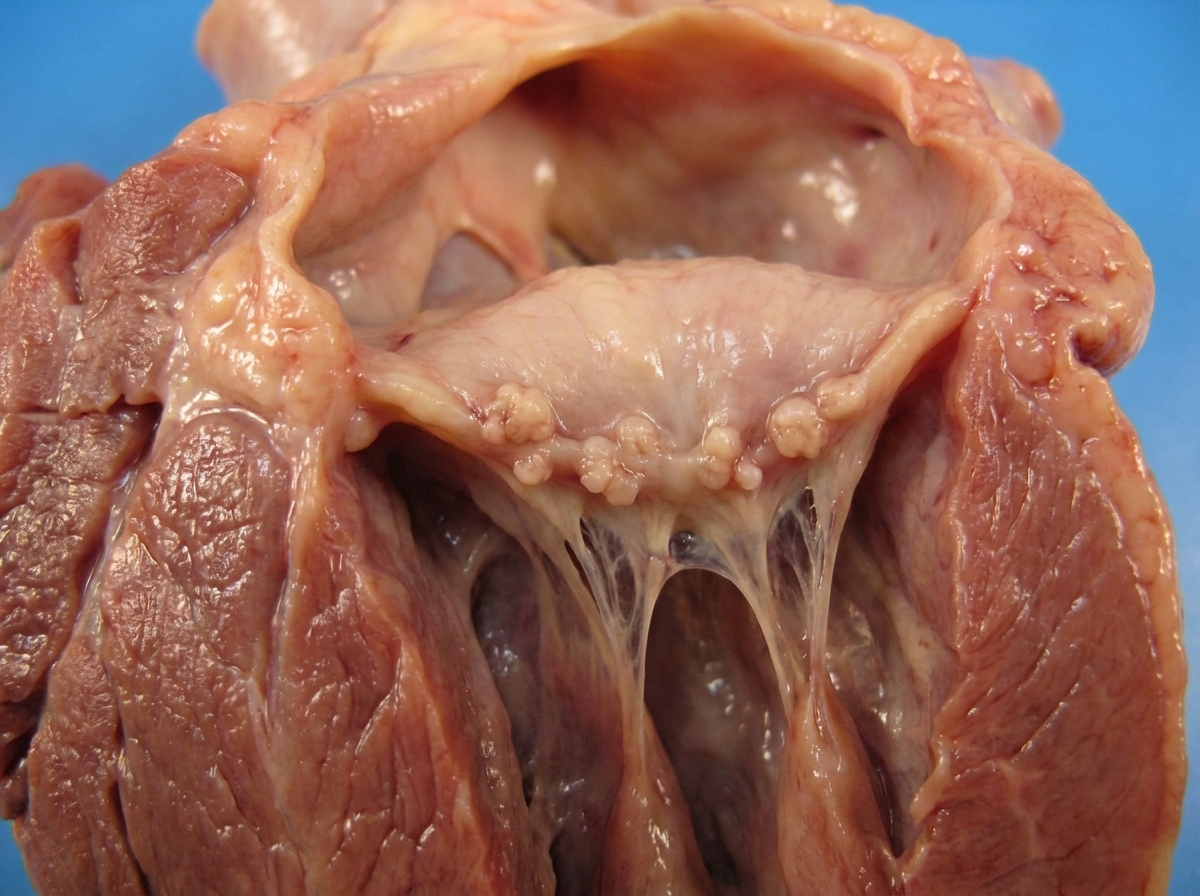
Gross findings of the heart from an 18-year-old female presented with a history of sore throat 3 months back and joint pains have been shown below. On auscultatory finding, a murmur was noted. What is your diagnosis?
What is the average time taken for a myocardial infarction to be completely healed?
A 36-year-old man with a history of long-standing rheumatic heart disease and mitral stenosis dies during cardiac surgery. Autopsy findings are consistent with mitral stenosis and reveal the presence of "heart failure cells." What process leads to the observation of "heart failure cells"?
At the time of autopsy of a 39-year-old female who died of complications of systemic lupus erythematosus, several medium-sized vegetations are found on both sides of the mitral valve and tricuspid valve. These cardiac vegetations are most likely the result of?
Practice by Chapter
Congenital Heart Disease
Practice Questions
Ischemic Heart Disease
Practice Questions
Hypertensive Heart Disease
Practice Questions
Valvular Heart Disease
Practice Questions
Myocarditis and Cardiomyopathies
Practice Questions
Pericardial Disease
Practice Questions
Cardiac Tumors
Practice Questions
Heart Failure Pathophysiology
Practice Questions
Cardiac Transplantation Pathology
Practice Questions
Endocarditis
Practice Questions
Want unlimited practice?
Get full access to all questions, explanations, and performance tracking.
Scan to download app
