Cardiac Pathology — MCQs

On this page
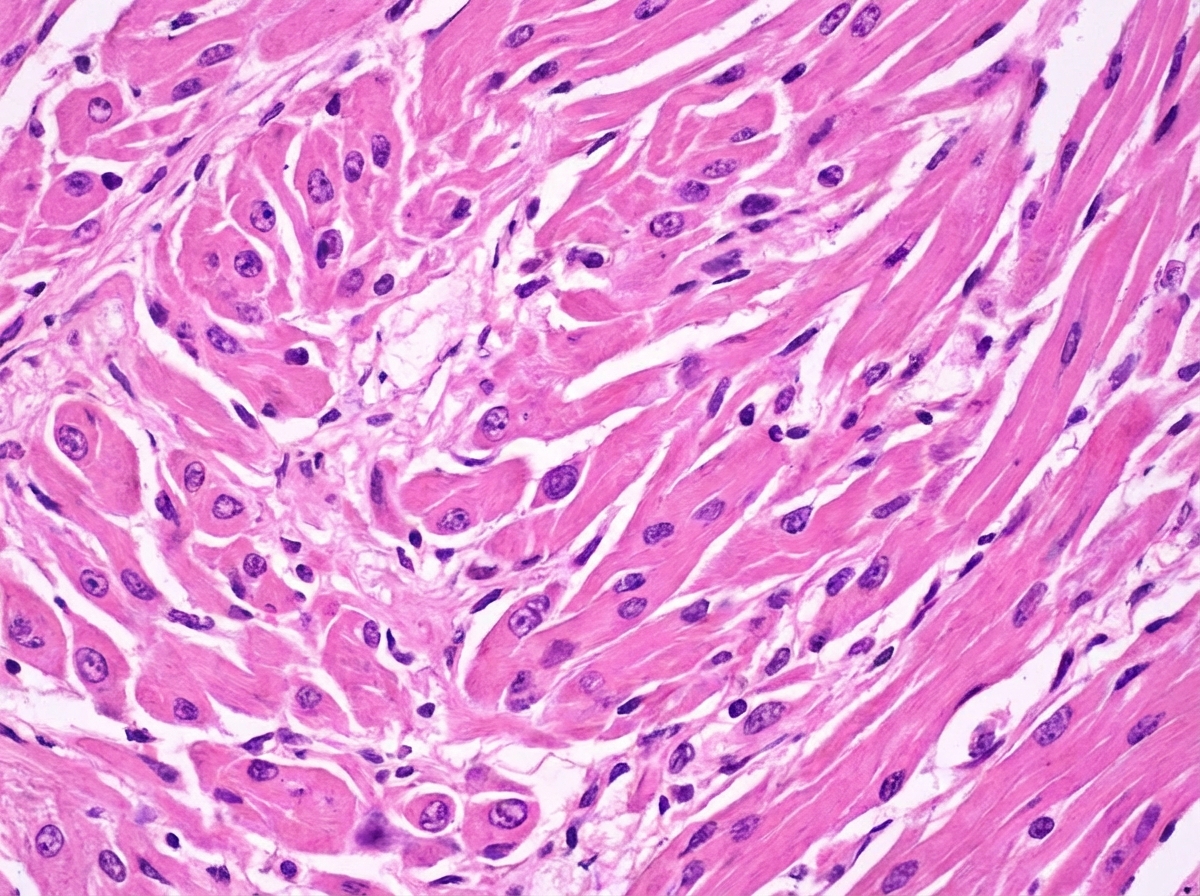
What type of cardiomyopathy is characterized by this arrangement of myofibrils?
All of the following statements regarding endocarditis are true, except:
What is the typical site of lesion in endocarditis of rheumatic heart disease?
Aschoff bodies in the myocardium are the hallmark of carditis associated with which condition?
Firm warty vegetations along the line of apposition of heart valves are present in which condition?
McCallum's patch is diagnostic of which condition?
A stenotic valve is removed from a 70-year-old man. The valve demonstrates hard nodular masses heaped up within the sinuses of Valsalva. On microscopic section, the acellular masses stain darkly blue with hematoxylin and eosin. Which of the following is the MOST significant constituent of the masses?
A patient developed an anterior wall myocardial infarction and died within 2 hours of symptom onset. An autopsy is being performed, and the involved myocardium is being observed under light microscopy. What pathological finding would be expected?
Aschoff's nodules are seen in which condition?
Non-sterile vegetations are seen in which of the following conditions?
Practice by Chapter
Congenital Heart Disease
Practice Questions
Ischemic Heart Disease
Practice Questions
Hypertensive Heart Disease
Practice Questions
Valvular Heart Disease
Practice Questions
Myocarditis and Cardiomyopathies
Practice Questions
Pericardial Disease
Practice Questions
Cardiac Tumors
Practice Questions
Heart Failure Pathophysiology
Practice Questions
Cardiac Transplantation Pathology
Practice Questions
Endocarditis
Practice Questions
Want unlimited practice?
Get full access to all questions, explanations, and performance tracking.
Scan to download app
