Neonatology and Perinatology — MCQs

On this page
Vaginal defense is lost:
Which of the following is NOT a common complication in a neonate born to a mother with diabetes mellitus?
What are the characteristics after 28 weeks of gestation?
Low birth weight baby is defined as a baby weighing:
A baby is born with a large defect in the occipital bone through which the posterior portion of the brain has herniated. Which of the following terms best describes this lesion?
During a clinical examination, a senior resident asks an intern to examine the umbilical cord. How many arteries and veins are normally present in a healthy umbilical cord?
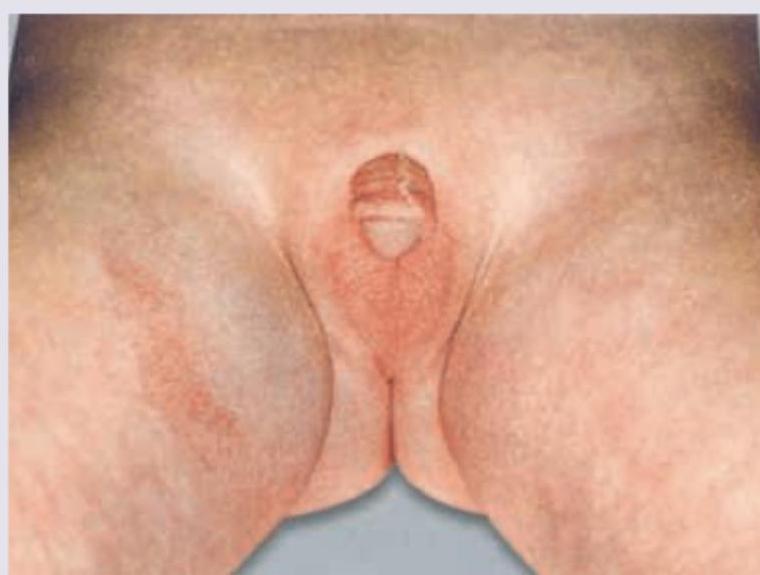
A Lethargic hypoglycemic girl neonate has the following genital appearance. What is the probable cause? (Recent Neet Pattern 2016-17)
Which of the following can be a complication in the baby due to post maturity of pregnancy ?
Meconium aspiration syndrome can be prevented by taking the following measures except :
A woman suffering from active tuberculosis not on ATT has a full term vaginal delivery. All the following should be done except:
Practice by Chapter
Neonatal Resuscitation
Practice Questions
Care of the Normal Newborn
Practice Questions
Low Birth Weight and Prematurity
Practice Questions
Neonatal Infections
Practice Questions
Birth Asphyxia and Hypoxic-Ischemic Encephalopathy
Practice Questions
Neonatal Jaundice
Practice Questions
Respiratory Distress in Newborn
Practice Questions
Congenital Anomalies
Practice Questions
Birth Injuries
Practice Questions
Perinatal Mortality and Morbidity
Practice Questions
Want unlimited practice?
Get full access to all questions, explanations, and performance tracking.
Scan to download app
