Contraception and Family Planning — MCQs

On this page
Which of the following is contained in the 'Today' contraceptive?
What is the ideal contraceptive in the given scenario?
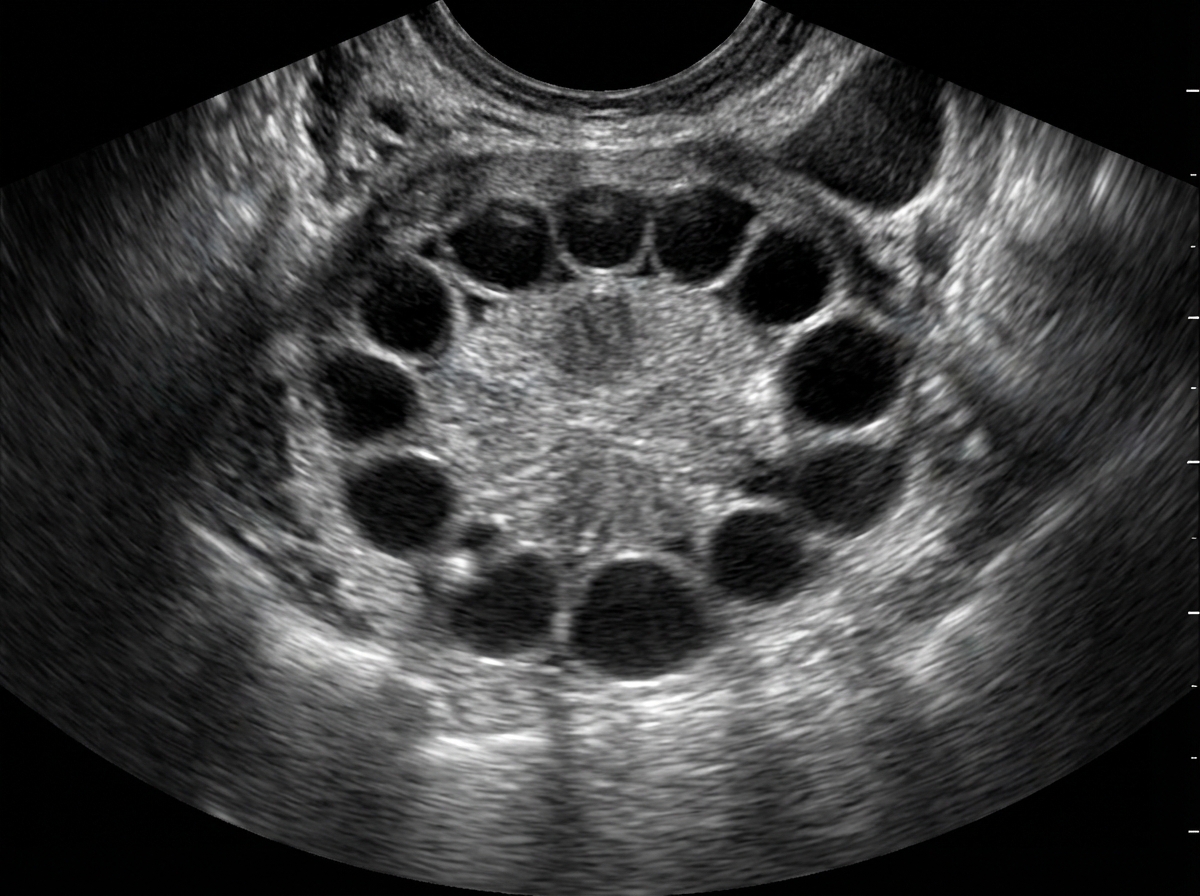
What is the absolute contraindication for prescribing oral contraceptive pills in a woman of reproductive age group?
Which is the most likely complication of an IUD?
Which of the following is a male contraceptive pill?
What is the primary mechanism of action of combined oral contraceptive pills?
Which method of contraception is contraindicated in a patient with AIDS?
Which of the following is an absolute contraindication for combined oral contraceptive pills?
Which one of the following statements regarding the female contraceptive "Today" is true?
Use of oral contraceptives decreases the incidence of all of the following except?
Practice by Chapter
Natural Family Planning Methods
Practice Questions
Barrier Methods
Practice Questions
Hormonal Contraceptives
Practice Questions
Intrauterine Devices
Practice Questions
Emergency Contraception
Practice Questions
Permanent Contraception Methods
Practice Questions
Contraception in Special Populations
Practice Questions
Contraceptive Counseling
Practice Questions
Side Effects and Complications of Contraceptives
Practice Questions
Future Contraceptive Technologies
Practice Questions
Want unlimited practice?
Get full access to all questions, explanations, and performance tracking.
Scan to download app
