Extracorporeal Removal Techniques — MCQs

Protective level of Tetanus anti-toxin is:
Following are used in treatment of digitalis toxicity except-
According to ATLS classification of hemorrhagic shock, a patient with decreased blood pressure, decreased urine output and decreased circulatory volume of 30-40% is managed by?
Drug X has an affinity for albumin, while drug Y has 150 times greater affinity. Which of the following statements is MOST accurate?
For toxicological analysis, which preservative is most appropriate for viscera?
After IV drug administration, elimination of a drug depends on:
Which of the following is not typically performed during septoplasty?
Which electrolyte imbalance causes prolonged QT interval?
A common finding in osteomalacia is
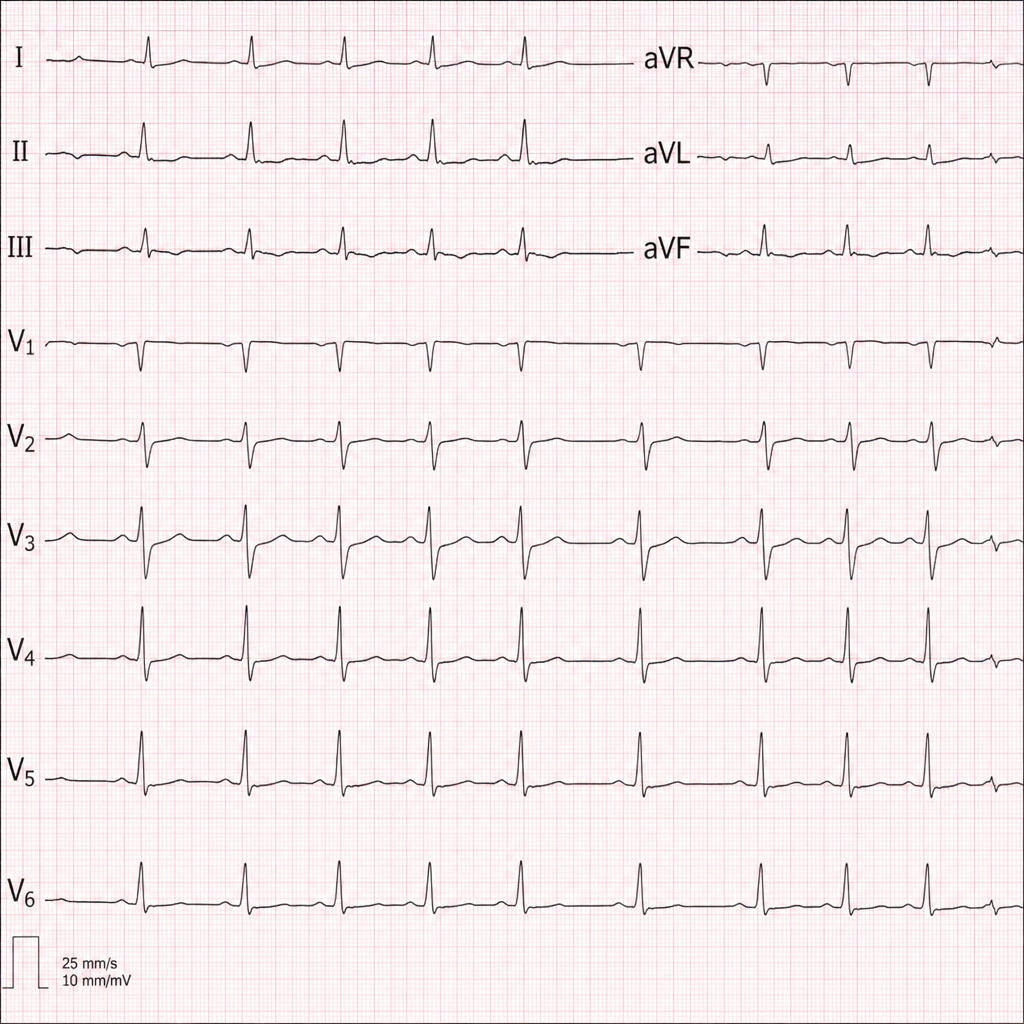
Diagnose the underlying medical disorder based on the ECG changes.
Want unlimited practice?
Get full access to all questions, explanations, and performance tracking.
Scan to download app
