Evidence-Based Medicine — MCQs

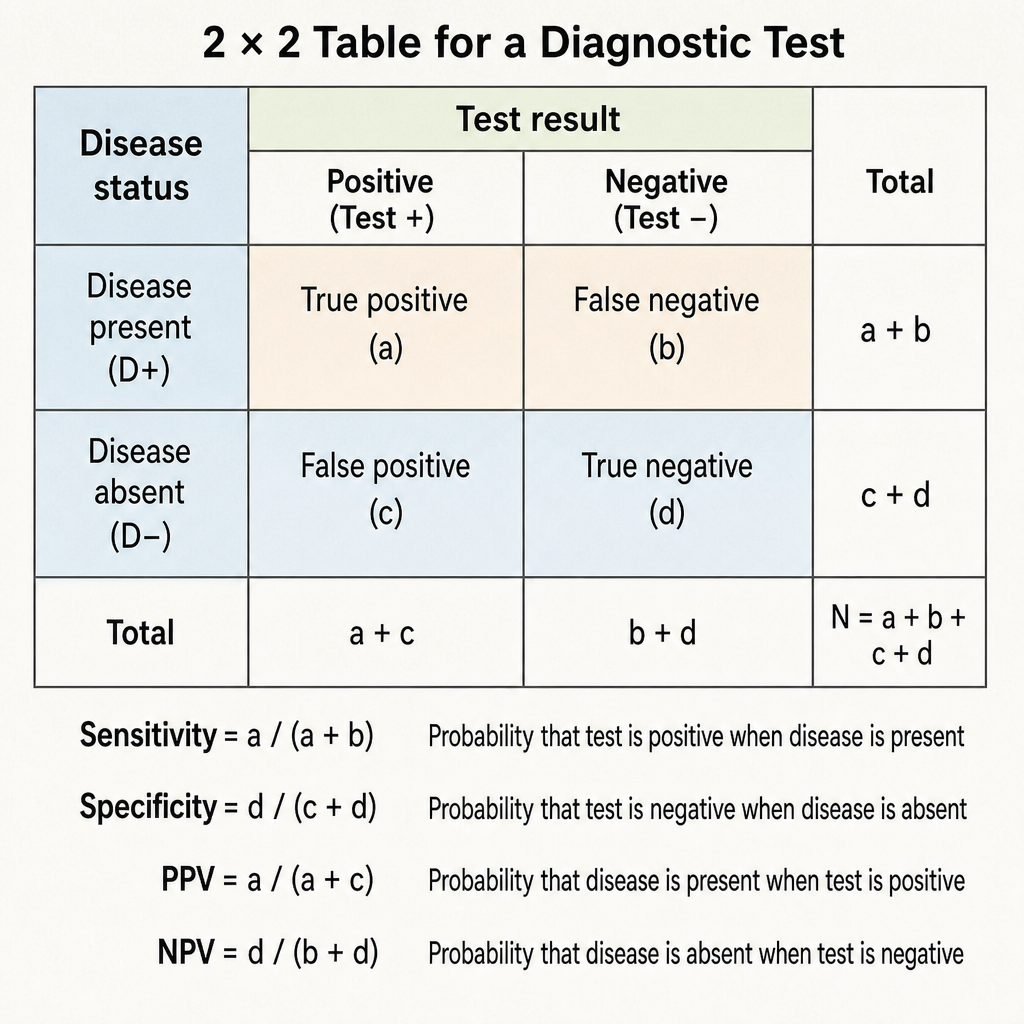
Calculate the sensitivity of a screening test: True Positives=80, False Negatives=20, True Negatives=90, False Positives=10
Specificity of a diagnostic test is defined as:
Which of the following is a type of observational study that analyzes population-level data?
Which of the following statements about screening for disease is false?
A study is to be conducted to compare the fat content in the expressed breast milk of pre-term infants with that of term infants. Which study design is best suited?
What type of evidence do medical certificates provide?
Which test is used for detecting gunshot residue?
Which of the following is not a characteristic of a systematic review?
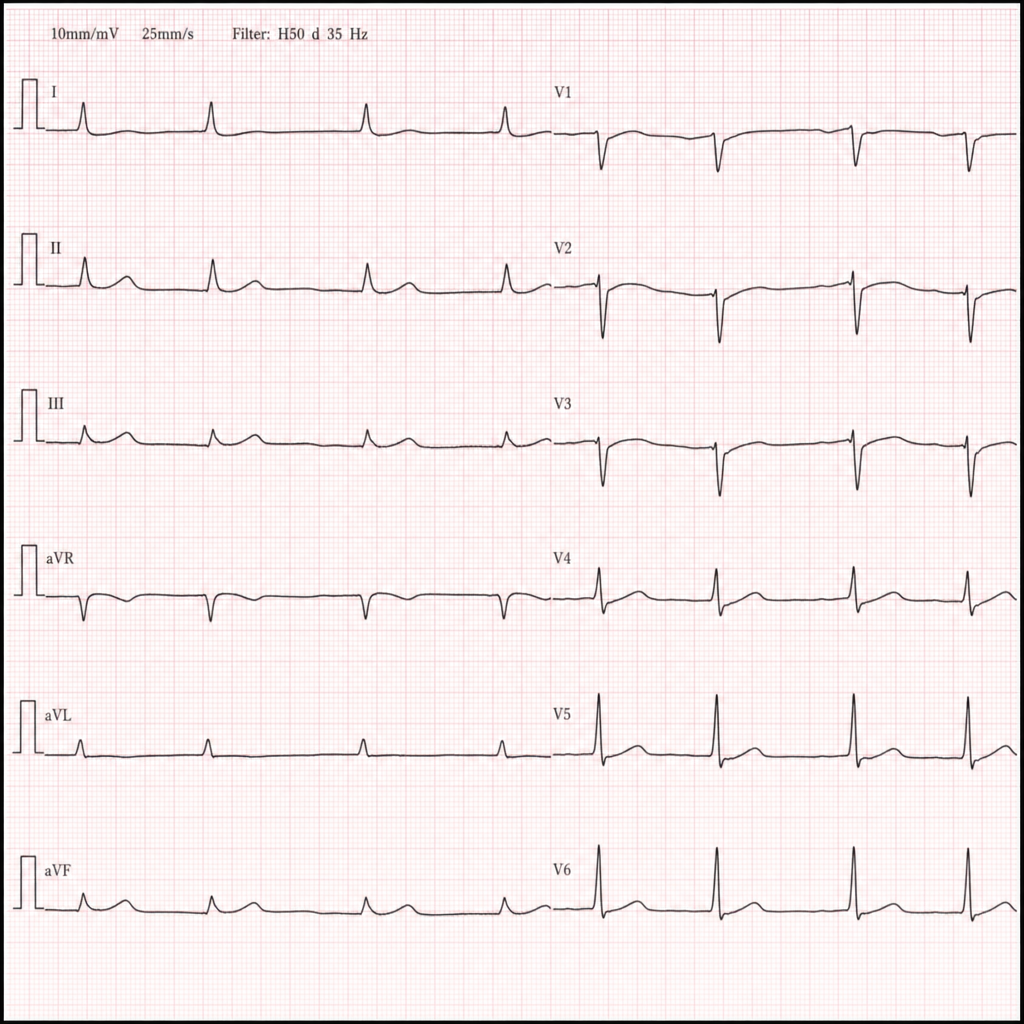
A homeless man is brought to the emergency department in January after being found unresponsive in a park. He is bradycardic and minimally responsive. A 12-lead ECG is obtained (Image 1). Which of the following is the most likely life-threatening arrhythmia if this patient's core temperature is not corrected?
A junior resident discovers that a senior consultant has been ordering unnecessary investigations for personal financial gain. After informal discussion is ignored, what is the most appropriate next step according to professional ethics?
Want unlimited practice?
Get full access to all questions, explanations, and performance tracking.
Scan to download app
