Oncology — MCQs

On this page
Alpha-fetoprotein (AFP) is a marker for which of the following conditions?
Maffucci syndrome is characterized by which of the following?
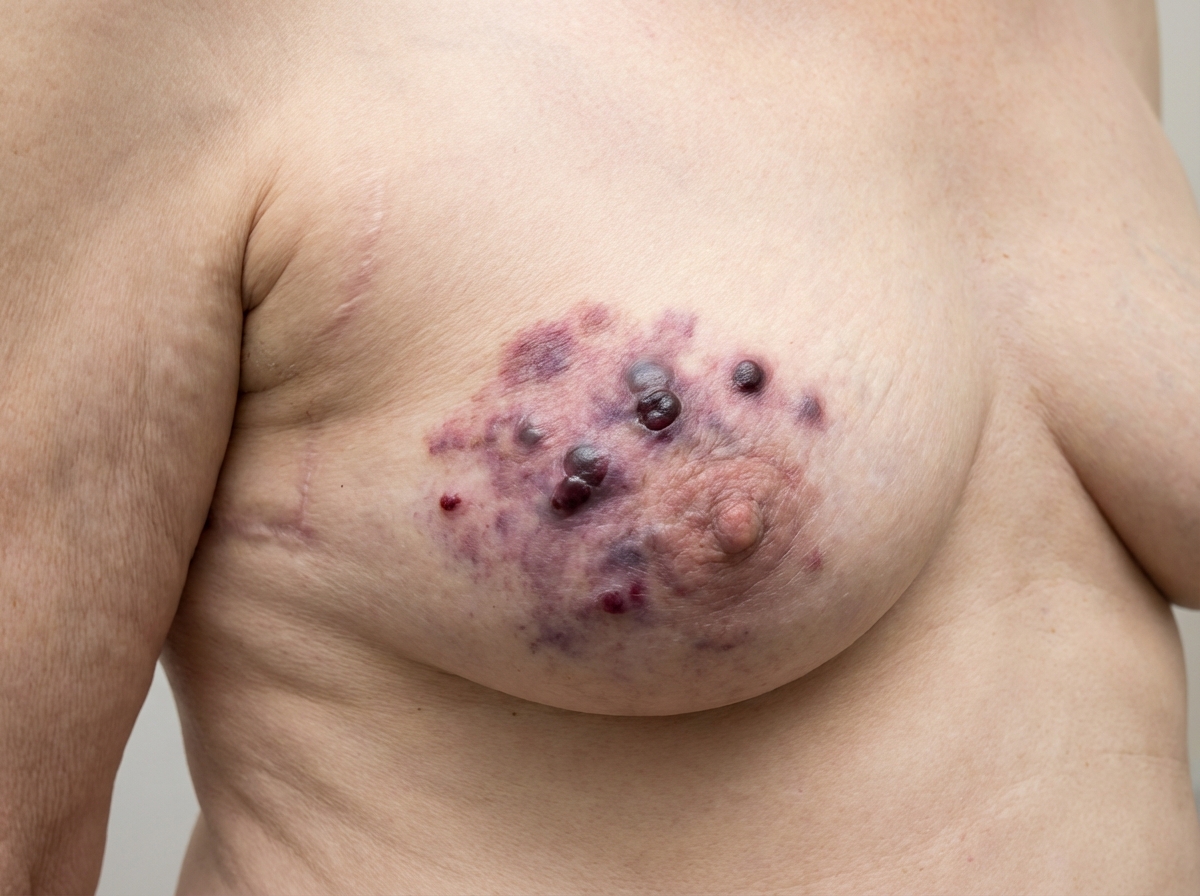
A 72-year-old woman presented with skin changes over her left breast. She had a history of breast cancer 5 years earlier and was treated with a lumpectomy and radiation therapy. What is the diagnosis?
What is the most common cause of ectopic ACTH production?
What is true about malignant melanoma?
What are the commonly used criteria for response evaluation during cancer treatment?
What is true about PEComa (perivascular epithelioid cell tumor)?
A patient with a Pancoast tumor develops a loss of voice after radiation. What is the most likely cause of this symptom?
Bilateral Renal cell carcinoma is seen in which of the following conditions?
Tumor lysis syndrome causes all of the following EXCEPT:
Practice by Chapter
Cancer Biology and Carcinogenesis
Practice Questions
Principles of Cancer Diagnosis and Staging
Practice Questions
Solid Tumor Management
Practice Questions
Hematological Malignancies
Practice Questions
Cancer Emergencies
Practice Questions
Principles of Chemotherapy, Immunotherapy and Targeted Therapy
Practice Questions
Radiation Oncology Basics
Practice Questions
Palliative Care in Oncology
Practice Questions
Cancer Screening and Prevention
Practice Questions
Paraneoplastic Syndromes
Practice Questions
Want unlimited practice?
Get full access to all questions, explanations, and performance tracking.
Scan to download app
