Hematological Malignancies — MCQs

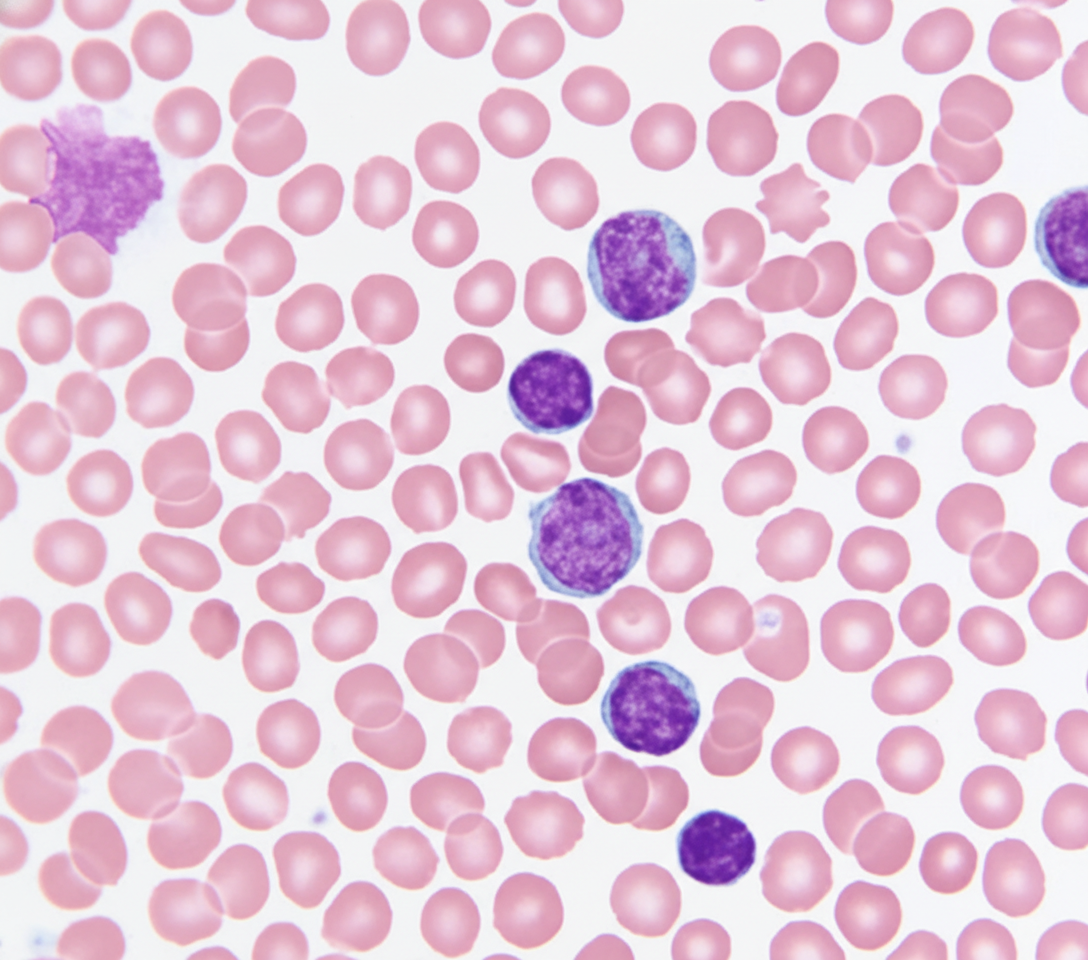
A 69-year-old Caucasian man presents for a routine health maintenance examination. He feels well. He has no significant past medical history. He takes aspirin for the occasional headaches that he has had for over several years. He exercises every day and does not smoke. His father was diagnosed with a hematologic malignancy at 79 years old. The patient’s vital signs are within normal limits. Physical examination shows no abnormalities. The laboratory test results are as follows: Hemoglobin 14.5 g/dL Leukocyte count 62,000/mm3 Platelet count 350,000/mm3 A peripheral blood smear is obtained (shown on the image). Which of the following best explains these findings?
A boy presents with fever, night sweats, and neck swelling. The biopsy of swelling showed a starry sky appearance. What is the most likely genetic abnormality seen in this case?
A 20-year-old male presented with fatigue, weakness, and jaundice. What is the most likely diagnosis?
Which of the following types of leukemia is administered prophylactic methotrexate for CNS prophylaxis –
In acute myeloid leukemia, Auer rods are numerous in which?
Multiple myeloma is characterized by which of the following?
Among the following AML subtypes, non-specific esterase (NSE) staining is typically NEGATIVE in which one?
The most common cause of malignant adrenal mass is:
Which of the following is true regarding carcinoid tumor?
Which of the following is the most common hematologic malignancy associated with Neurofibromatosis-1 (NF-1) in a child?
Want unlimited practice?
Get full access to all questions, explanations, and performance tracking.
Scan to download app
