Lipid Disorders — MCQs

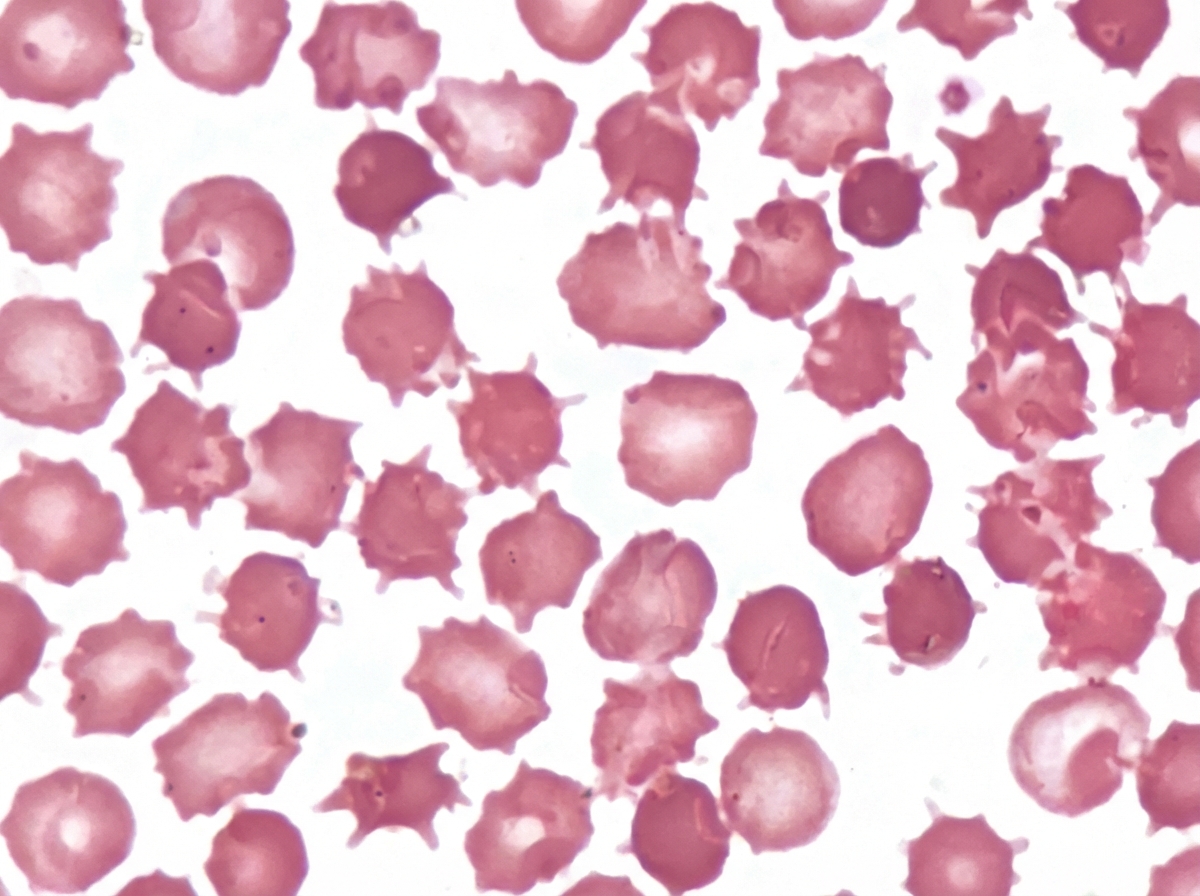
An adult male presented with a protruding abdomen, diarrhea, visual symptoms, and neurological manifestations. His LDL is low. Based on the peripheral smear finding shown in the image, what is the likely diagnosis?
A person is diagnosed with familial type IIa hyperlipoproteinemia. What is the basic defect in this type of hyperlipoproteinemia?
Which apolipoprotein is the primary structural component of LpA-I particles?
Which of the following enzymes is not targeted by hypolipidemic drugs?
What is the drug with the highest efficacy to increase plasma HDL?
Which of the following represents a desired lipid parameter for cardiovascular risk control in hypertension?
All of the following are true about nephrotic syndrome except?
HMG-CoA reductase is inhibited by:
Vitamin B12 deficiency can give rise to all of the following, except which of the following?
Which of these conditions is classified as a nephritic syndrome?
Want unlimited practice?
Get full access to all questions, explanations, and performance tracking.
Scan to download app
