Drug Development and Clinical Trials — MCQs

Which study design is most effective for investigating rare adverse effects of a drug?
Which one of the following drugs has a narrow therapeutic range?
Which of the following statements about phase IV clinical trials is correct?
Match the following drugs in Column A with their contraindications in Column B. | Column A | Column B | | :-- | :-- | | 1. Morphine | 1. QT prolongation | | 2. Amiodarone | 2. Thromboembolism | | 3. Vigabatrin | 3. Pregnancy | | 4. Estrogen preparations | 4. Head injury |
What is the primary purpose of interventional studies in clinical research?
Declaration of Oslo deals with:
What is a key benefit of Randomized Controlled Trials (RCTs) in clinical research?
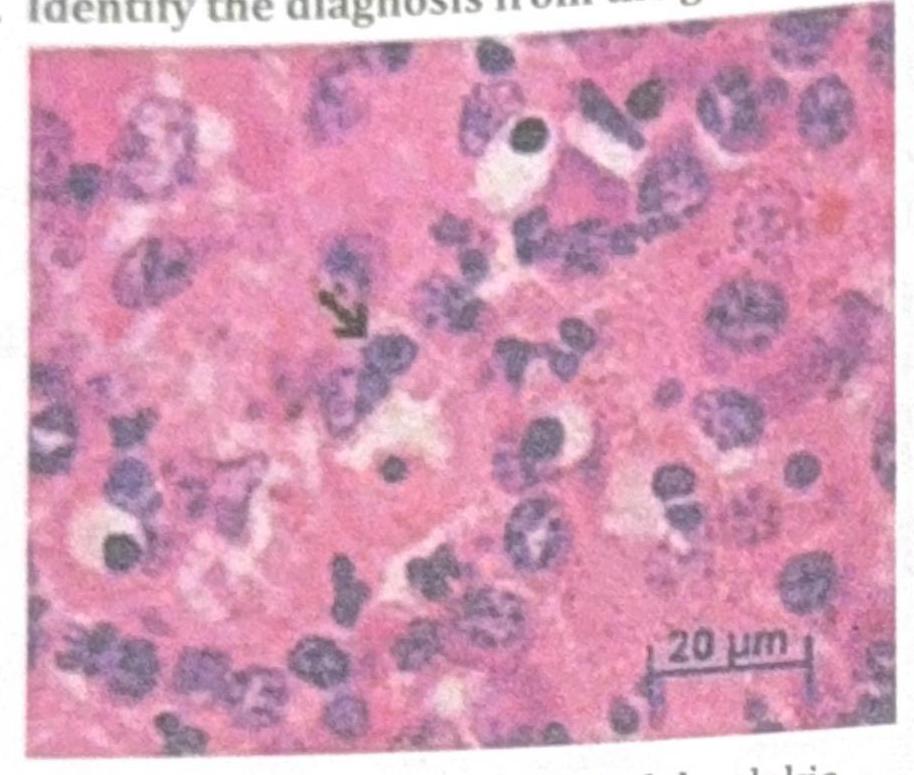
Identify the diagnosis from the given image
Which of the following is an example of placebo?
Which blinding technique is considered the most effective in clinical trials?
Want unlimited practice?
Get full access to all questions, explanations, and performance tracking.
Scan to download app
