Clinical Pharmacology — MCQs

On this page
A diabetic female on Isoniazid (INH) and Rifampicin for tuberculosis developed Deep Vein Thrombosis (DVT). She was started on Warfarin, but her Prothrombin Time (PT) is not elevated. What is the next appropriate step in management?
Which antibiotic is MOST frequently implicated as a cause of drug-induced liver injury (DILI)?
Among the drugs used for Hepatitis B antiviral therapy, which of the following has the highest tendency for peripheral neuropathy?
Which of the following drugs should not be given in renal failure?
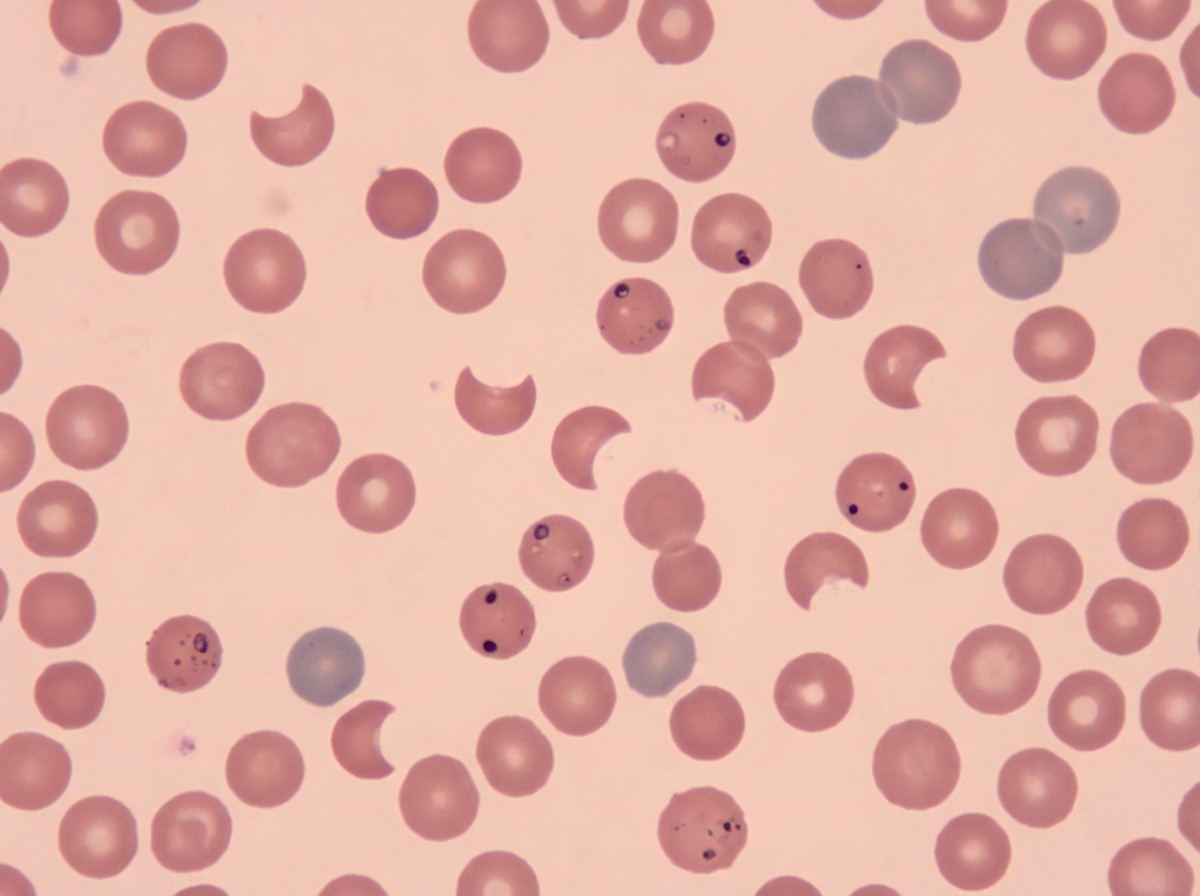
A 30-year-old male presented with a skin lesion in his right axilla. A presumptive diagnosis of staphylococcal skin carbuncle was made and the patient was treated with empiric trimethoprim/sulfamethoxazole. After 2 days, the patient presented to the emergency department with excessive weakness, abdominal pain, and dark-colored urine. On examination, vital signs were normal, and jaundice was present. Laboratory studies showed a drop in Hb from 14 g/dl to 8 g/dl and a rise in bilirubin levels. Urine dipstick was positive for bilirubin. Peripheral blood smear findings are suggestive of a specific condition. What is the most likely diagnosis for this patient?
Which metabolic abnormality is caused by cyclosporine?
A 40-year-old lady presents to casualty with abdominal pain and vomiting. She also reports hearing people conspiring to harm her, and visual hallucinations. These symptoms started along with the abdominal pain following the use of barbiturates. What is the most likely diagnosis?
Which of the following drugs is most commonly associated with noninfectious chronic meningitis due to drug hypersensitivity?
Which of the following antitubercular drug combinations is preferred in patients with severe liver disease?
All of the following cause hypertension except?
Practice by Chapter
Pharmacokinetics and Pharmacodynamics
Practice Questions
Drug Metabolism and Excretion
Practice Questions
Adverse Drug Reactions and Interactions
Practice Questions
Therapeutic Drug Monitoring
Practice Questions
Pharmacogenomics and Precision Medicine
Practice Questions
Drug Development and Clinical Trials
Practice Questions
Rational Prescribing and Deprescribing
Practice Questions
Medication Adherence Strategies
Practice Questions
Special Populations (Pediatric, Geriatric, Pregnancy)
Practice Questions
Pharmacovigilance
Practice Questions
Want unlimited practice?
Get full access to all questions, explanations, and performance tracking.
Scan to download app
