Immunization and Vaccine-Preventable Diseases — MCQs

On this page
How many vaccine vials can a carrier transport daily?
Which of the following vaccines does not provide herd immunity?
What is the recommended minimum age for vaccination with the human papillomavirus (HPV) vaccine?
What is the recommended vaccination strategy for rubella?
The influenza vaccine contains all strains of influenza virus except which one?
All of the following is true about Hepatitis B Vaccination except?
How much time does it take for the finding shown below, after BCG vaccination?
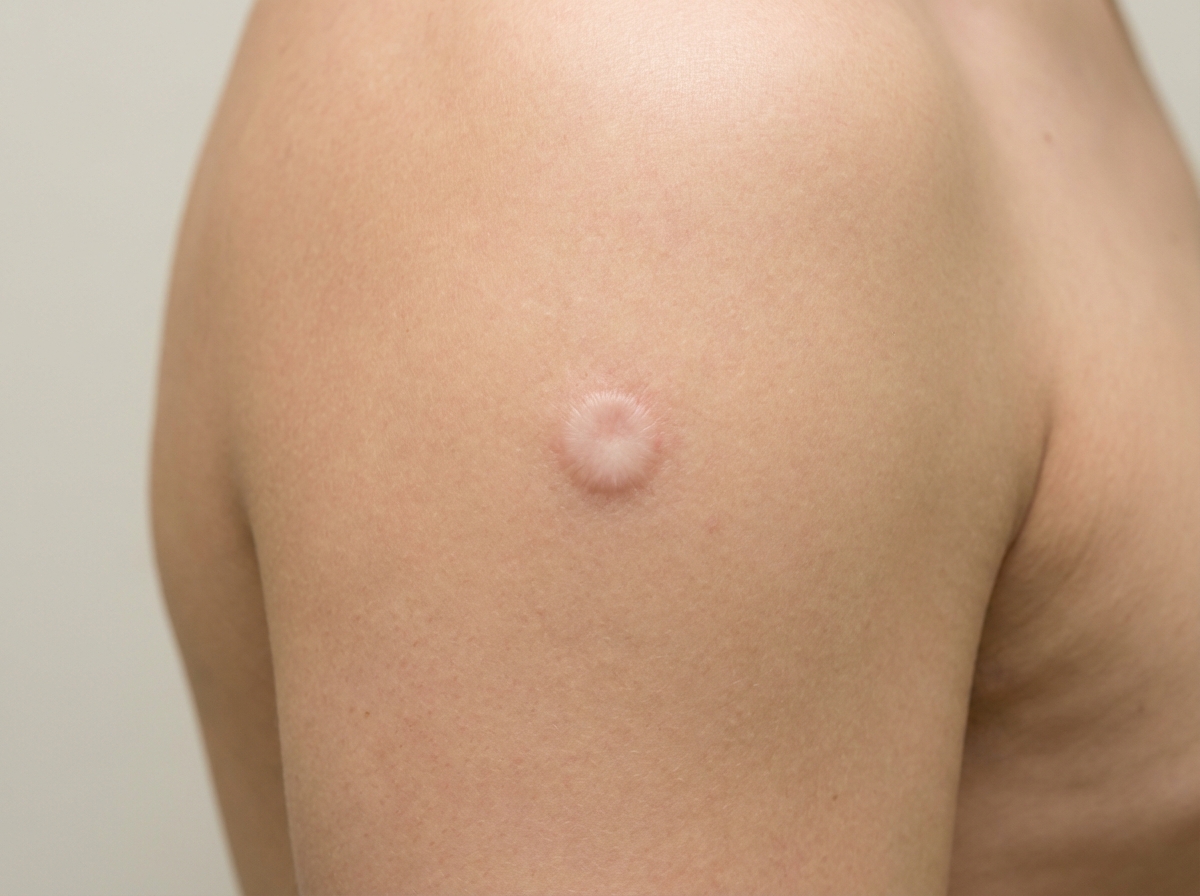
What immunization is typically given at 9 months of age?
What is true about the DPT vaccine?
All of the following are contraindications to pertussis-containing vaccines EXCEPT?
Practice by Chapter
Principles of Immunization
Practice Questions
Types of Vaccines
Practice Questions
Universal Immunization Program
Practice Questions
Cold Chain System
Practice Questions
Vaccine Storage and Handling
Practice Questions
Adverse Events Following Immunization
Practice Questions
National Immunization Schedule
Practice Questions
Polio Eradication
Practice Questions
Measles Elimination
Practice Questions
Tetanus Control
Practice Questions
New and Underutilized Vaccines
Practice Questions
Vaccination Coverage Assessment
Practice Questions
Want unlimited practice?
Get full access to all questions, explanations, and performance tracking.
Scan to download app
