Immunization and Vaccine-Preventable Diseases — MCQs

On this page
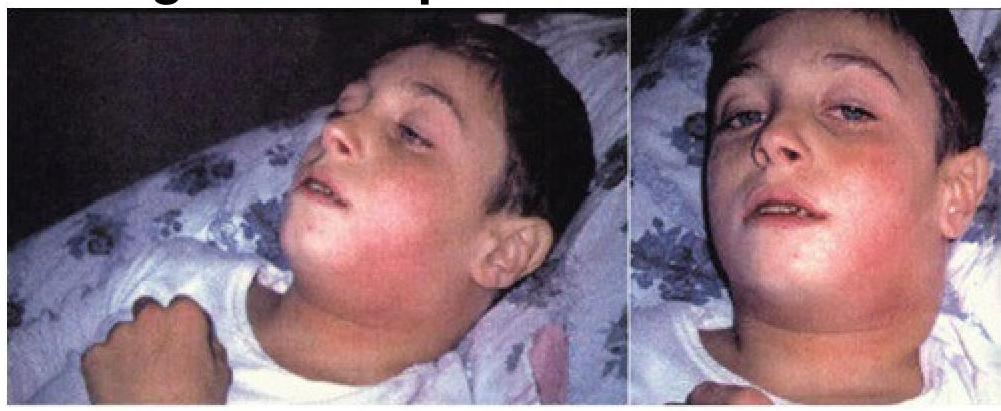
A patient with the following feature shown in the image. The patient reports having another 3-year-old sibling at home, who is fully immunized as per the immunization schedule. What is the best measure to prevent diphtheria in the sibling of the child with diphtheria?
Which vaccine is not included in the Indradhanush mission across all states?
Which of the following statements about the MR vaccination campaign launched by WHO is false?
Japanese encephalitis vaccine in routine schedule is given in how many doses -
Which vaccine is the most widely used globally in childhood vaccination programs, aside from the Oral Polio Vaccine (OPV)?
Vaccine derived polio virus outbreaks are due to?
Which Diphtheria vaccine is recommended for a 14-year-old girl?
Which of the following statements regarding polio vaccination is false?
Immunity starts after how many days of yellow fever vaccination ?
Which of the following statements about the Pneumococcal Polysaccharide Vaccine (PPV) is correct?
Practice by Chapter
Principles of Immunization
Practice Questions
Types of Vaccines
Practice Questions
Universal Immunization Program
Practice Questions
Cold Chain System
Practice Questions
Vaccine Storage and Handling
Practice Questions
Adverse Events Following Immunization
Practice Questions
National Immunization Schedule
Practice Questions
Polio Eradication
Practice Questions
Measles Elimination
Practice Questions
Tetanus Control
Practice Questions
New and Underutilized Vaccines
Practice Questions
Vaccination Coverage Assessment
Practice Questions
Want unlimited practice?
Get full access to all questions, explanations, and performance tracking.
Scan to download app
