Immunization and Vaccine-Preventable Diseases — MCQs

On this page
A pregnant woman with a 2-year-old child gives a history of completing required antenatal vaccinations during her previous pregnancy. Which of the following would you recommend for her current pregnancy?
Which of the following vials, as shown in the image, can be used for administering vaccines?
Identify person shown in the image below:

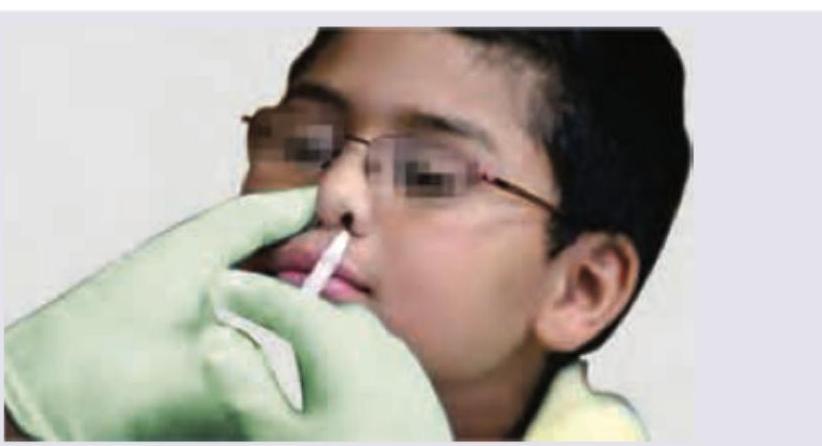
All are correct regarding the following route of vaccine administration except:
All are correct about the vaccine shown except:

Which is correct about the Vaccine Vial monitor shown in the image? (Recent NEET Pattern 2016-17)

All are correct about Mission Indradhanush (the launched programme) except:
Consider the following statements with regard to Ice-Lined Refrigerator (ILR) employed for storing vaccines at the sub-district level : I. These types of refrigerators are top-opening. II. Based on the temperature zone, the inside of ILRs can be divided into 3 parts. III. The upper part of ILR is cooler compared to the lower part. IV. Vaccines should never be kept directly on the floor of the ILR as they can get damaged. Which of the statements given above are correct?
In a child who has not received any dose of DPT and OPV immunization, up to what age can these vaccines be given under the Universal Immunization Programme?
Which among the following vaccines best represent interventions that focus on cancer prevention too ? 1. Hepatitis A vaccine 2. Hepatitis B vaccine 3. Hemophilus influenzae B vaccine 4. Human papilloma virus vaccine Select the correct answer using the code given below :
Practice by Chapter
Principles of Immunization
Practice Questions
Types of Vaccines
Practice Questions
Universal Immunization Program
Practice Questions
Cold Chain System
Practice Questions
Vaccine Storage and Handling
Practice Questions
Adverse Events Following Immunization
Practice Questions
National Immunization Schedule
Practice Questions
Polio Eradication
Practice Questions
Measles Elimination
Practice Questions
Tetanus Control
Practice Questions
New and Underutilized Vaccines
Practice Questions
Vaccination Coverage Assessment
Practice Questions
Want unlimited practice?
Get full access to all questions, explanations, and performance tracking.
Scan to download app
