Principles of Epidemiology — MCQs

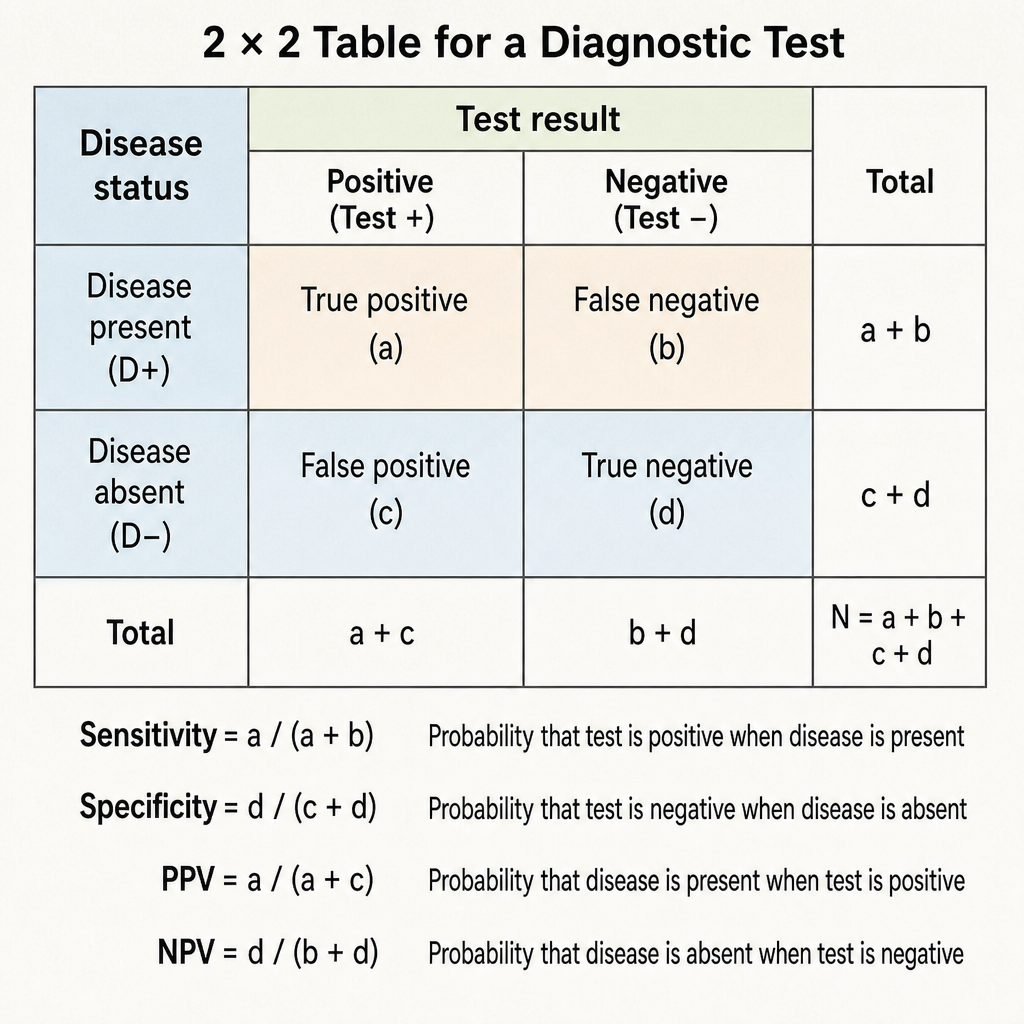
Specificity of a diagnostic test is defined as:
Which of the following is not considered a type of subject bias?
Which of the following best reflects the diagnostic power of a test?
Which one of the following is a good index of the severity of an acute disease?
According to Hill's criteria, which of the following is NOT a criterion for establishing causality in noncommunicable diseases?
Which type of epidemiological study uses a population as the unit of study?
What is the primary purpose of interventional studies in clinical research?
Descriptive epidemiology studies the distribution and determinants of health-related states or events in specified populations. Which of the following best describes the fundamental components of descriptive epidemiology?
The "risk of a disease" is measured by the
What is the death rate among cholera-affected individuals in a population of 5000, where 50 people are affected by cholera, and 10 of these individuals have died?
Want unlimited practice?
Get full access to all questions, explanations, and performance tracking.
Scan to download app
